Credit: KS Kim/iStock/Getty Images Plus
It’s well-established that cell-free protein synthesis (CFPS) is a highly efficient way to produce complex therapeutic proteins and that vesicle delivery systems are very effective transport systems. Now, researchers report that synergies caused by combining those two technologies have “significant potential” to advance biomanufacturing by enhancing both production efficiency and complex protein delivery.
“This powerful synergy simultaneously streamlines biotherapeutic production and enhances therapeutic outcomes through improved protein stability, increased bioavailability, and precise spatiotemporal control of drug delivery,” according to researchers Wonhee Kim, PhD, professor; Jinjoo Han, doctoral student; Shraddha Chauhan, PhD, postdoctoral researcher; and Jeong Wook Lee, PhD, associate professor, all at Pohang University of Science and Technology in Korea, writing in a recent paper.
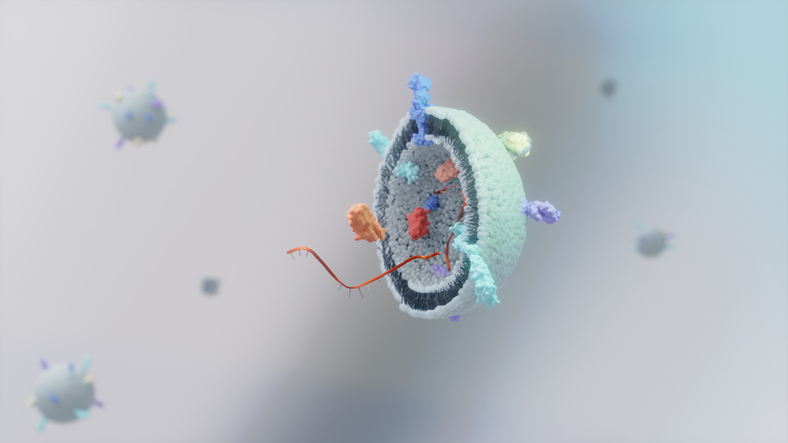
Specifically, integrating CFPS and vesicle-based delivery enhances membrane proteins’ stability and functionality. That’s because they are produced in a lipid bilayer that resembles the conditions present during natural, physiological production. The precisely controlled manipulations that are possible in this in vitro system have helped scientists successfully complete such challenging endeavors as overexpressing G protein-coupled receptors and such recombinant membrane proteins as outer membrane porin F.
“Eukaryotic-based CFPS systems containing endoplasmic reticulum-derived vesicles have the potential for a wide range of biotechnological applications,” Kim and colleagues note. “Those systems enable post-translational modifications,” and, as others have shown, glycosylation, which allows more diverse glycoproteins to be synthesized… and thus “expand the spectrum of expressible proteins.” Adding “sensing and producing programs… and…more complex genetic networks…provides further refinement in therapeutic control,” they report.
For CFPS, cell free vesicles (CFVs) such as liposomes, polymersomes, and microsomes have several advantages that extracellular vesicles (EVs) lack. Their encapsulation efficiency rate is nearly 100%—nearly twice that of EVs. Also, they “enable streamlined development and rapid prototyping,” thanks to in vitro optimization that enables high-throughput functional testing without the complexity of cell line engineering. Separate purification of biologics from cells or media isn’t needed, and the systems are scalable, they note.
There are challenges, however—namely, the low efficiency of CFPS systems, the inability to accurately predict the behavior of CFV genetic systems in vivo, imperfect targeting, and potential immunogenicity of CFVs.
When selecting CFVs, Kim and colleagues advise considering some of the newer alternatives, too. Dencrimersomes, for example, have uniform particle sizes and better membrane permeability than liposomes. Another option, EV-liposome hybrid vesicles, offers the targeting attributes of EVs and the encapsulation efficiency, loading capacity, and engineering flexibility of liposomes.
“CFPS systems derived from host-compatible microbes or EV-liposome hybrids offer a promising strategy to reduce CFV-associated immune responses,” the team concludes. Nonetheless, they caution, “Intensive research is (still) required.”