Scientists from University of California, Los Angeles, Harvard University, and elsewhere have developed what they claim is the first comprehensive road map of how the ovarian reserve forms in primates. Insights from their work could lead to new treatments for female infertility and hormonal disorders such as polycystic ovary syndrome (PCOS). Full details are published in a new Nature Communications paper titled “Defining the cell and molecular origins of the primate ovarian reserve.”
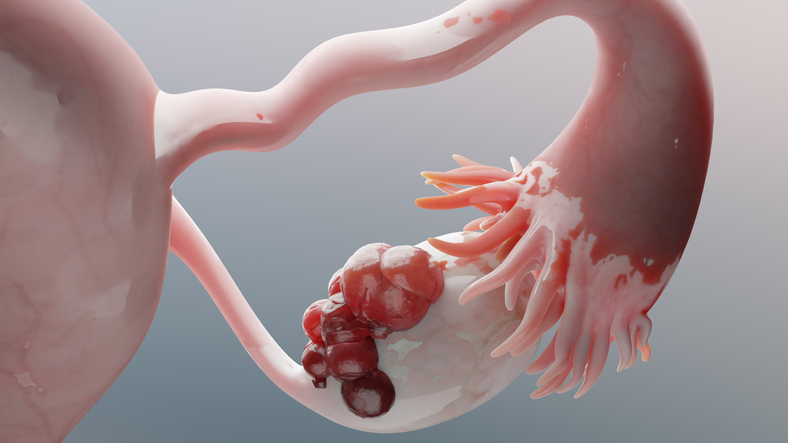
By way of background, the ovarian reserve refers to the lifetime supply of eggs that females are born with. It serves not only as the foundation for reproduction but also drives hormone production in the ovaries. According to Amander Clark, PhD, a professor of molecular, cell, and developmental biology and a member of UCLA’s Broad Stem Cell Research Center, the scientists’ map offers “a manual” to “help scientists create more accurate human ovarian models to better study ovarian disease and dysfunction.”
Despite its role in reproductive development, crucial aspects of how the ovarian reserve is built has largely remained a mystery. One of the challenges of studying the process in humans is that the ovarian reserve forms entirely in utero, which is a challenging window of time to study. To overcome this obstacle, the research team used rhesus macaques. The primate shares about 93 percent of its DNA with humans and undergoes similar ovarian and ovarian reserve development, making it an ideal physiological model.
Their first step in the study was to identify critical stages in ovarian reserve development including initial ovary formation, female sex determination, and follicle formation—the process by which protective sacs develop around eggs in the ovaries to support their survival. Next they used single-cell sequencing and spatial transcriptomics to analyze these developmental snapshots at the cellular and molecular level.
A key finding from their analysis was a cellular explanation for mini-puberty, which refers to an unexplained hormone surge that occurs soon after a baby’s birth. The scientists observed that specialized hormone-producing cells activate in the ovary shortly before birth. This period of so-called “practice growth” is responsible for the hormone spike detected during mini-puberty. The absence of this hormone surge could serve as an early biomarker for ovarian dysfunction disorders like PCOS. Insights like this matter because “if we can identify risk factors in infancy that impact ovarian health, then early interventions can be made so that these women don’t suffer once they go through puberty,” Clark said.
Among other benefits, access to this data contained in the atlas has immediate applications for stem cell researchers who have long been interested in growing accurate ovarian organoids in the lab for fertility studies and other use cases. In fact, Clark and her team are already working on generating ovarian support cells from induced pluripotent stem cells. They can combine these with lab-grown germ cells to generate three-dimensional ovarian models.
“Women’s health is already understudied, but the ovary in particular has been neglected,” said Sissy Wamaitha, PhD, a postdoctoral scholar in the Clark lab and first author on the paper. “To effectively treat reproductive health conditions—as well as the growing number of general health issues we now acknowledge affect people with ovaries—we must first develop a fundamental understanding of the full scope of this organ’s function.”