In a groundbreaking discovery poised to reshape the therapeutic landscape for head and neck cancers linked to Human Papillomavirus (HPV), researchers from Henry Ford Health and Michigan State University Health Sciences have identified a pivotal mechanism cancers employ to evade immune detection. Their work, recently published in the prestigious Proceedings of the National Academy of Sciences (PNAS), illuminates how the protein MARCHF8 actively dismantles MHC-I molecules on cancer cells, effectively enabling tumors to hide from immune surveillance. This understanding opens transformative avenues in cancer immunotherapy, promising renewed hope for patients grappling with notoriously resistant malignancies.
HPV-positive head and neck squamous cell carcinomas have alarmingly surged in incidence across the United States over the past several decades. These tumors starkly differ from other cancer types in their capacity to suppress surface expression of MHC class I (MHC-I) molecules—critical immune markers that signal cellular distress and trigger immune responses. The absence of MHC-I severely hampers the immune system’s ability to recognize and target cancer cells, rendering the tumors effectively “invisible” to cytotoxic T lymphocytes and natural killer (NK) cells. Until now, the biochemical machinery facilitating this immune evasion remained enigmatic, hampering efforts to devise effective immune-based therapies.
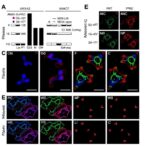
The investigative team, led by Dohun Pyeon, Ph.D., a professor specializing in Microbiology, Genetics, and Immunology, uncovered that the viral-driven upregulation of the membrane-associated E3 ubiquitin ligase MARCHF8 is at the heart of this immunological stealth. MARCHF8 tags MHC-I molecules on the cancer cell surface with ubiquitin, marking them for degradation through the cellular proteasomal system. This targeted destruction prevents MHC-I molecules from presenting tumor-associated antigens to immune cells, thereby incapacitating the host’s natural defense mechanisms.
Experimental models where the researchers genetically knocked out MARCHF8 yielded remarkable results. The restoration of MHC-I surface expression abruptly reactivated immune recognition. CD8+ T cells and NK cells, critical effectors of anti-tumor immunity, infiltrated the tumor microenvironment in force, orchestrating potent and coordinated cytotoxic responses against the previously shielded cancer cells. Notably, this immune resurgence converted immunologically “cold” tumors—those refractory to existing immunotherapies—into “hot” tumors amenable to immune attack, highlighting the therapeutic potential of targeting MARCHF8.
Mohamed Khalil, Ph.D., the study’s first author, emphasized the dual benefit of disrupting MARCHF8: “Our data show that knocking out MARCHF8 not only suppresses tumor growth directly but also invigorates the immune system’s ability to identify and eliminate cancer cells by enhancing the infiltration and activation of T cells, NK cells, and macrophages.” This multifaceted boosting of the tumor immune microenvironment is crucial since the immunosuppressive milieu is a major barrier in effective cancer treatment.
Integral to deciphering the cellular complexity within tumors, the collaborative effort with Dr. Qing-Sheng Mi employed state-of-the-art single-cell RNA sequencing technologies. This strategy revealed that loss of MARCHF8 fundamentally reprograms intercellular communication within the tumor microenvironment, significantly amplifying the cytotoxic functionalities of immune effector cells. Such high-resolution insights clarify the mechanistic basis behind the immune reactivation and will underpin the development of precision therapies.
The potential clinical ramifications of this discovery are profound. By developing pharmacological inhibitors of MARCHF8, physicians could restore MHC-I expression on tumor cells in patients, rendering their cancers once again visible to the immune system. The envisioned therapeutic paradigm involves combining MARCHF8 blockade with current immunotherapeutic agents, such as checkpoint inhibitors, to synergistically induce tumor regression. This approach aims to provide a desperately needed lifeline to patients whose cancers have thus far defied conventional immune-based treatments.
While the immediate findings focus on HPV-positive head and neck cancers, the implications could extend broadly across oncology, given that immune evasion via MHC-I downregulation is a strategy employed by diverse tumor types. Continued research will explore the nuanced roles of different immune cells, including natural killer cells, whose newfound prominence in this context challenges prior assumptions and suggests additional targets for therapeutic intervention.
Supported by a $3 million grant from the National Institute of Dental and Craniofacial Research, along with strategic funding from the MSU Foundation and the Henry Ford + MSU Cancer Seed Funding Program, the team’s efforts are advancing rapidly toward translational applications. The next steps involve screening and optimizing MARCHF8 inhibitors and evaluating their efficacy and safety in preclinical models before progressing to human clinical trials.
According to Professor Pyeon, “Our research not only demystifies a critical cancer immune escape mechanism but also sparks new possibilities to fundamentally alter treatment outcomes. By preventing tumors from shredding their red flags, we can empower the immune system to do what it does best—eradicate malignancies.” This landmark study signifies a paradigm shift in understanding tumor immunology and exemplifies the power of cross-disciplinary collaboration in tackling some of the most challenging cancers of our time.
As the landscape of cancer treatment increasingly pivots toward harnessing the patient’s own immune system, discoveries such as the role of MARCHF8 in immune evasion are essential. They bridge gaps between molecular cancer biology and clinical application, setting the stage for innovative therapies that can overcome resistance and improve survival rates for patients worldwide.
Subject of Research: Cells
Article Title: The membrane-associated ubiquitin ligase MARCHF8 degrades MHC-I in HPV-positive head and neck cancer for immune evasion
News Publication Date: March 9, 2026
Web References: https://www.pnas.org/doi/10.1073/pnas.2525730123
Image Credits: Debbie Walton, Michigan State University Department of Microbiology, Genetics, & Immunology
Keywords: Cancer, Immunology, HPV, Head and Neck Cancer, MARCHF8, MHC-I, Immune Evasion, Tumor Microenvironment, Immunotherapy
Tags: Cancer immunotherapy strategiescytotoxic T lymphocyte evasionHPV-associated squamous cell carcinoma researchHPV-positive head and neck cancersHPV-related cancer immune suppressionimmune evasion mechanisms in cancerImmune Surveillance in CancerMARCHF8 protein role in cancerMHC class I downregulation in tumorsmolecular targets for HPV-positive cancersnatural killer cell immune escapetherapeutic approaches for resistant cancers