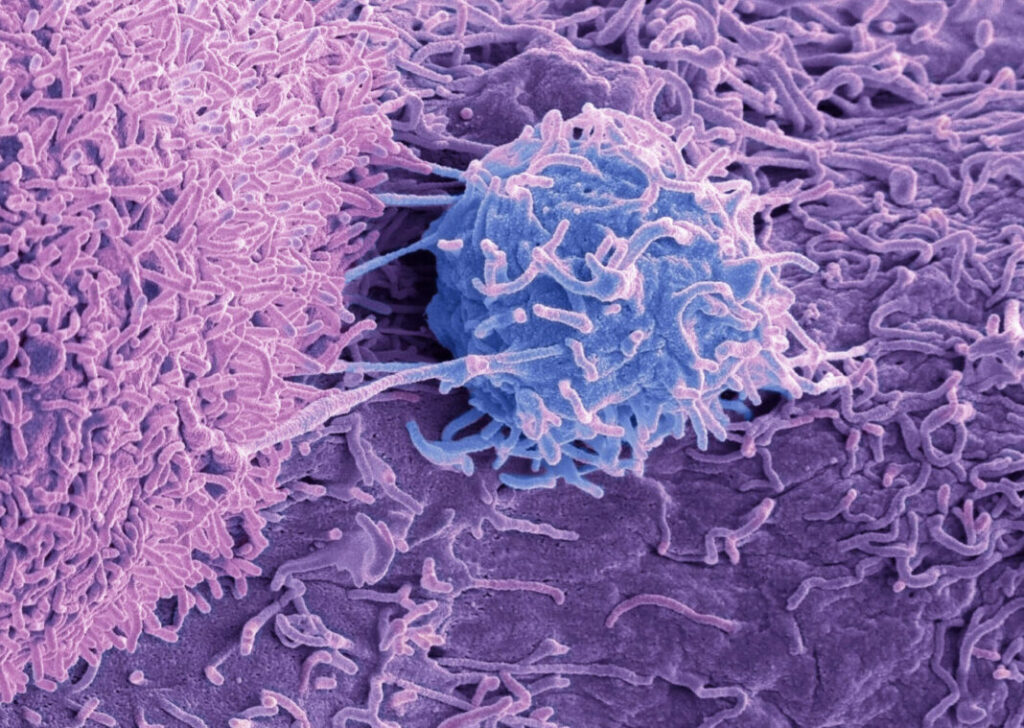
Much of the challenge in developing cancer therapies is determining methods to improve the effectiveness of the immune system’s ability to identify and kill cancer cells. While therapies like CAR T and check point inhibitors help in cancer treatment, they have their limitations.
A cross-country research group from the University of California, Los Angeles (UCLA), Stanford School of Medicine, University of Utah, and Columbia University have engineered T cells to be more efficient and precise in their ability to kill prostate cancer cells. The technique involves a modification to cellular receptors in T cells, which naturally create a weak “fishhook-like” interaction. Their work strengthens the bond between the T cell and a target tumor cell, improving the cytotoxic function of the T cell.
“In our collaborative work, we demonstrate that just a single amino acid change introducing these ‘fishhooks’ is sufficient to transform immune cells into a potent killer mode,” said study co-senior author K. Christopher Garcia, PhD, from the Stanford School of Medicine.
The work was published in a paper entitled, “Overcoming T cell tolerance to tumor self-antigens through catch-bond engineering,” in Science.
T cells form mechanical bonds with targets—catch bonds—which allow them to sense and respond to threats. The team found that modifying just two amino acids in the T cell receptor allowed these bonds to last longer, improving the T cell’s ability to recognize tumor targets.
“By engineering catch bonds, we aim to benefit more patients by overcoming immune tolerance,” said co-senior author Owen N. Witte, MD, of the UCLA Health Jonsson Comprehensive Cancer Center.
The researchers engineered multiple versions of T cell receptors (TCR). They focused specifically on “a weakly reactive TCR specific for a non-mutated human prostate tumor associated antigen, Prostatic Acid Phosphatase.” After engineering multiple variants of TCR156, two versions were identified as highly effective at recognizing tumors, releasing cancer-killing molecules, proliferating, and resisting exhaustion.
“Using advanced structural studies at atomic resolution, we were able to demonstrate how a tiny change, just one amino acid in the interface between a T cell receptor and a prostate cancer protein called PAP, can extend the bond lifetime, dramatically boosting the T cell’s ability to kill tumors in living models,” said co-first author Xiaojing Tina Chen, PhD, a postdoctoral researcher at the Stanford School of Medicine.
These modified T cells underwent confirmation analysis, including advanced imaging, single-cell RNA sequencing, and structural analysis. Further, the team conducted modeling analyses to show that the mutations didn’t change the overall function of the TCR shape.
Functionality experiments showed that the engineered TCR 156 bonded with PAP for longer periods of time, and the T cells showed increased secretion of tumor-killing molecules, including Granzyme B, IFNγ, and TNFα. On a larger scale, using mouse models, they found that the engineered T cells were able to delay or stop tumor growth, while control T cells had little effect on the tumor growth.
“This work shows that tumor control can be linked to a single molecular bond,” said co–first author Zhiyuan Mao, PhD, a postdoctoral researcher at UCLA.
Engineered T cells also persisted longer, maintained a stem-like state, and resisted exhaustion compared with unaltered T cells—a common limitation of other T cell therapies.
“By creating T cells that are stronger, longer-lasting, and highly precise, the approach moves the field closer to safer and more effective adoptive cell therapies for patients,” said Witte.
This study demonstrates the potential for TCR therapy to be effective in clinical settings. Although native TCR binding is weak, small modifications can have a big impact on functionality, and therapy effectiveness.
“These findings suggest that catch bond engineering could be a generalizable strategy to enhance T cell therapies for many cancers,” concluded Garcia.