Sponsored content brought to you by
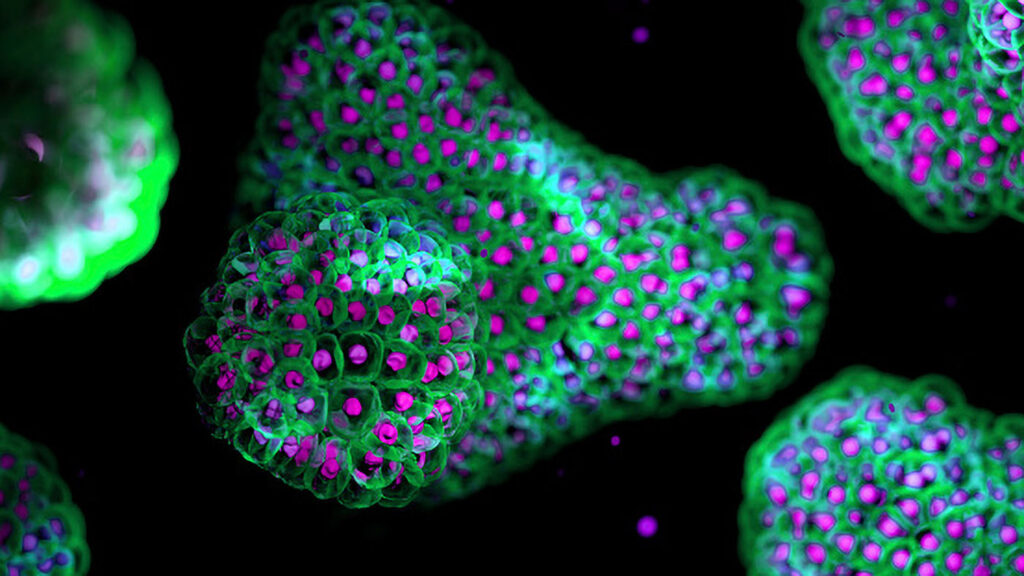
Drug discovery increasingly relies on three-dimensional (3D) cell models to address a persistent challenge in biomedical research: traditional experimental systems often fail to accurately reflect human biology. Organoids, spheroids, and other complex cellular models allow scientists to recreate aspects of tissue structure and function in the laboratory. These systems now play a central role in new approach methodologies (NAMs), experimental strategies designed to produce human-relevant biological data while reducing reliance on animal testing.
“Some of the most interesting applications of 3D models are rare disease and complex-disease modeling,” says Robert Padilla, field application scientist at Corning Life Sciences. “With historical models we often lack biological relevancy, and animal models don’t always recapitulate human biology the way we want.”
That disconnect between animal studies and human biology has long slowed drug development. Compounds that perform well in animals frequently fail during clinical trials, forcing researchers to spend additional time and resources identifying therapies that actually work in people. Three-dimensional cell models aim to narrow that gap by recreating tissue-like environments where cells interact with neighboring cells and the surrounding extracellular matrix.
“With the regulatory environment opening up for NAMs, we can generate human-relevant data faster,” Padilla explains. “And we don’t have to rely on costly animal models as the starting point.”
Keep it consistent
Early in his career, Padilla was taking a cardiomyocyte process from 2D into 3D development. “One of the biggest challenges was generating spheroids with uniform size,” he says.
Uniform spheroids matter because stem-cell differentiation relies on tightly controlled signaling environments. Differences in spheroid size can change how cytokines and growth factors penetrate the tissue, producing cells that mature at different rates.
“When I generated spheroids that varied even within 25% of each other, they matured at different time points,” Padilla says. “That meant I didn’t have a fully controlled process.”
Technologies that generate uniform spheroids therefore improve experimental reproducibility and enable more reliable screening assays. Corning recently introduced its Elplasia technology, which organizes single cells into uniform aggregates using microcavities. “You can generate extremely uniform spheroids across the entire plate,” Padilla says. Moreover, a new Elplasia-based vessel can generate up to 48,000 spheroids in a single container. “Now we’re generating the numbers of cells researchers actually need for therapy development,” Padilla adds.
Expanding applications
Producing thousands of organoids, however, creates new analytical challenges. High-throughput imaging technologies allow scientists to simultaneously monitor large numbers of spheroids. Automated microscopes and phenotypic screening platforms capture images and molecular data from each tissue model, enabling researchers to evaluate drug responses across massive datasets.
“When we generate tens of thousands of spheroids, we need technologies that can analyze all of them,” Padilla explains.
Corning’s products and analytical capabilities support one of the most promising applications of NAMs: modeling complex physiological barriers. One example involves the blood–brain barrier, which blocks most drugs from reaching brain tissue.
Padilla highlights a screening system that combines 3D–glioma tumor spheroids with laboratory models of the blood–brain barrier using Corning’s Transwell plates. Researchers can test whether candidate therapies cross the barrier and kill tumor cells.
“In one screen, we started with 27 potential drug candidates,” Padilla says. “Using this NAM approach, we reduced that list to nine.” Such a reduction dramatically simplifies drug development. By narrowing the field early, researchers can focus resources on the most promising compounds and accelerate progress toward clinical testing.
Padilla believes the field remains in its early stages but sees enormous promise ahead. By improving human relevance and experimental scalability, organoids and NAM platforms could fundamentally reshape how scientists study disease and develop new therapies. As he says, “We want to increase human relevancy and reduce the time it takes to bring therapies to market.”

Learn more www.corning.com.