In a groundbreaking study published in Pediatric Research, scientists have unveiled new insights into the complex interplay between mechanical stretch, Piezo ion channels, and store-operated calcium entry (SOCE) in the development of human airway smooth muscle. This discovery sheds light on fundamental physiological mechanisms that could reshape our understanding of respiratory health and disease, especially in early life. It also opens new therapeutic avenues for conditions characterized by dysfunctional airway smooth muscle behavior, including asthma and other obstructive airway diseases.
Human airway smooth muscle (ASM) plays a critical role in the regulation of airway tone and respiratory function. Unlike skeletal muscle, ASM is highly sensitive to mechanical forces and biochemical signals, making it a dynamic tissue capable of adjusting airway caliber in response to various stimuli. One of the key drivers of ASM function is calcium signaling. Intracellular calcium flux controls contraction and relaxation, dictating airway resistance and airflow. However, the precise molecular machinery that links mechanical cues to calcium dynamics during human airway development has remained elusive—until now.
The study focused on Piezo channels, which are mechanosensitive ion channels that transduce mechanical stimuli into calcium influx and other ion movements. Piezo1 and Piezo2, the two main family members, are widely expressed in various cells and tissues, serving as crucial sensors for stretch, pressure, and shear stress. While Piezo channels have been extensively studied in vascular endothelium and sensory neurons, their roles in developing ASM had not been thoroughly characterized prior to this investigation.
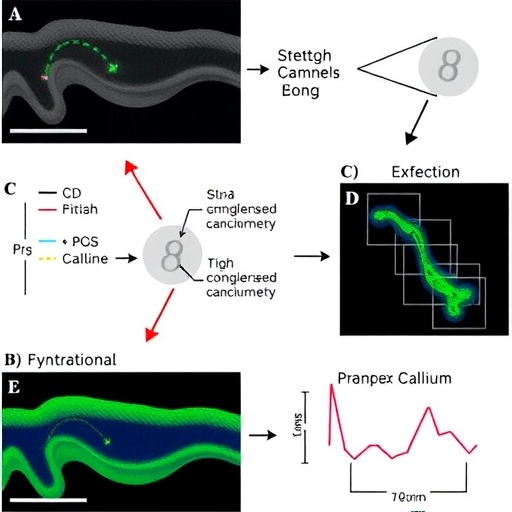
Using cutting-edge molecular biology techniques and functional assays on human ASM cells isolated from developing lungs, the researchers meticulously examined the expression pattern and activation profile of Piezo channels in response to controlled mechanical stretch. Their data revealed that exposing ASM cells to a physiologically relevant stretch stimulus robustly activated Piezo-mediated calcium influx. This activation was finely tuned, meaning that partial stretch induced moderate calcium entry, whereas sustained or excessive stretch drove higher influx, suggesting a graded response suitable for nuanced control of ASM function.
Crucially, the study also explored the relationship between Piezo channel activation and store-operated calcium entry (SOCE), a well-known calcium entry mechanism triggered by the depletion of calcium stores within the endoplasmic reticulum. SOCE involves the coordinated action of STIM and Orai proteins to replenish intracellular calcium, maintaining cellular calcium homeostasis and function. The researchers found a novel crosstalk wherein stretch-induced activation of Piezo channels modulated SOCE activity in developing human ASM cells. This unexpected link underscores a complex signaling network integrating mechanical stimuli with intracellular calcium regulation.
Further investigations showed that blocking Piezo channels pharmacologically or via gene silencing disrupted the normal calcium signaling pattern in response to stretch. This disruption led to diminished SOCE and altered ASM contractility, implying that Piezo channels are indispensable for proper mechanotransduction and calcium homeostasis in developing airways. These findings carry profound implications for understanding how airway smooth muscle develops functional properties during prenatal and early postnatal stages.
The implications of this research extend beyond basic biology. Airway smooth muscle hyperactivity and remodeling are hallmarks of pediatric respiratory diseases such as asthma and bronchopulmonary dysplasia. By elucidating the molecular pathways that regulate ASM calcium signaling during development, this study identifies potential molecular targets for early intervention. Therapeutic modulation of Piezo channels or SOCE components could fine-tune ASM responses, preventing or mitigating airflow obstruction caused by abnormal muscle contractility.
Moreover, the discovery that mechanical stretch, a naturally occurring physiological phenomenon during fetal breathing movements and postnatal respiratory effort, directly influences calcium entry via Piezo channels and SOCE adds a new dimension to developmental biology. It suggests that mechanical forces are not mere physical factors but essential biochemical regulators that shape airway structure and function from the earliest stages of life. This paradigm shift emphasizes the need to consider biomechanical environments in tissue engineering, regenerative medicine, and disease modeling.
The experimental design incorporated state-of-the-art calcium imaging, patch-clamp electrophysiology, and molecular interference techniques to rigorously dissect the roles of Piezo and SOCE pathways. High-resolution live-cell imaging demonstrated real-time calcium dynamics in ASM subjected to mechanical stretch, directly linking mechanical inputs with intracellular signaling events. Concurrent electrophysiological recordings validated the ion channel activity corresponding to the observed calcium influx.
On a cellular level, the interplay between Piezo channels and SOCE creates a feedback system that stabilizes intracellular calcium concentrations within a functional range. This balance prevents calcium overload, which could otherwise lead to cytotoxicity or dysregulated muscle contraction. Understanding these protective mechanisms has relevance for pharmacology, as excessive activation or inhibition of ion channels can have unintended consequences.
From a translational perspective, the findings pave the way for developing novel diagnostic tools and personalized therapeutic strategies. For example, biomarkers related to Piezo channel expression or function might predict susceptibility to airway hyperresponsiveness or guide dosing of calcium-modulating drugs. Future clinical trials could target these pathways to determine efficacy in managing pediatric airway diseases.
The study’s insights also prompt a re-examination of existing models of airway development and disease pathogenesis. Traditional views have often focused on inflammatory or genetic factors; however, this research highlights the integral role of mechanotransduction and ion channel biology. Integrating mechanical and biochemical signals into a holistic framework will enhance predictive modeling and improve identification of intervention points.
Furthermore, the researchers discussed potential environmental influences on Piezo channel activity during development. Factors such as prenatal exposure to hypoxia, toxins, or infections could alter mechanical signaling pathways, contributing to developmental airway disorders. Unraveling these complex interactions requires interdisciplinary approaches combining molecular biology, biomechanics, and clinical research.
In conclusion, this landmark study uncovers a crucial mechanistic link between mechanical stretch, Piezo ion channels, and store-operated calcium entry in developing human airway smooth muscle. By revealing how these pathways converge to regulate calcium signaling and muscle function, the research not only advances basic science but also holds promise for transformative clinical applications. As respiratory diseases continue to pose significant global health challenges, understanding the foundational biology governing airway physiology is more urgent than ever. This work represents a significant step forward in that journey.
As we look ahead, expanding investigations into the role of Piezo channels and SOCE in adult airway smooth muscle, as well as their involvement in disease states, will be critical. Harnessing this knowledge will undoubtedly open new frontiers in respiratory medicine, ultimately improving patient outcomes by targeting the roots of airway dysfunction at the molecular and mechanical levels.
Subject of Research: Mechanotransduction and calcium signaling pathways in developing human airway smooth muscle.
Article Title: Stretch, Piezo channels, and store operated calcium entry in developing human airway smooth muscle.
Article References:
Pfeffer-Kleemann, D.A., Thompson, M.A., Borkar, N.A. et al. Stretch, Piezo channels, and store operated calcium entry in developing human airway smooth muscle. Pediatr Res (2026). https://doi.org/10.1038/s41390-026-05006-3
Image Credits: AI Generated
DOI: 10.1038/s41390-026-05006-3 (24 April 2026)
Tags: airway smooth muscle contraction regulationcalcium entry in airway developmentcalcium signaling in airway smooth musclehuman airway smooth muscle physiologymechanical stretch and calcium influxmechanotransduction in respiratory healthobstructive airway disease mechanismspediatric respiratory muscle developmentPiezo ion channels in airway smooth musclestore-operated calcium entry mechanismsstretch-activated Piezo channelstherapeutic targets for asthma