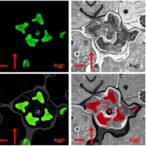
In a groundbreaking leap for cellular biology and microscopy, researchers at the Indian Institute of Science (IISc) have developed an enhanced DNA-PAINT imaging technique to visualize multiple biomolecules simultaneously within the nucleus of cancer cells at unprecedented resolution. This advancement opens a new frontier in understanding cellular nuclear architecture by unveiling the spatial organization of critical proteins involved in transcription machinery and nuclear structural integrity, providing one of the most detailed molecular maps of nuclear organization to date.
Cells, the building blocks of life, contain a complex milieu of biomolecules such as proteins and nucleic acids, orchestrating essential biological functions. Traditional microscopy techniques, limited by optical diffraction, have allowed imaging only a handful of molecular species simultaneously, typically two or three. This constraint has restricted researchers’ capability to untangle the intricate interactions and spatial arrangements of molecules essential for genomic regulation and cellular health.
Addressing this challenge, the IISc team employed DNA-PAINT (Points Accumulation for Imaging in Nanoscale Topography), a super-resolution microscopy technique that transcends conventional limits by exploiting the transient binding of fluorescently tagged DNA oligonucleotides to complementary DNA sequences tethered to biomolecules of interest. This method generates blinking fluorescent signals, allowing the precise localization of individual molecules with nanoscale accuracy.
What sets this research apart is the creation of an expanded repertoire of DNA tags, enabling the simultaneous targeting of up to 12 distinct biomolecular species within intact cells. The team engineered five of these probes to possess enhanced binding kinetics — binding faster and remaining attached longer — which significantly improves image quality and spatial resolution down to 3-5 nanometers. Such fidelity in imaging allows researchers to distinguish molecular arrangements at near-molecular scales, previously unattainable by optical methods.
Importantly, the strongly binding DNA tags reduce photodamage by requiring lower laser intensities during imaging, thus preserving the integrity of both the fluorescent probes and the delicate cellular structures. Photobleaching and molecular disruption, common pitfalls in fluorescence microscopy, are minimized, enabling prolonged imaging sessions without compromising data quality.
In addition to improving tagging chemistry, the IISc researchers optimized imaging protocols to accelerate data acquisition. Early implementations of DNA-PAINT required many hours to image a single molecular species due to the need for extensive sampling to achieve high localization precision. The enhanced technique now facilitates rapid, multiplexed imaging of nine different nuclear targets within a timeframe of less than four hours, marking a substantial improvement in experimental throughput.
This high-speed, multi-target imaging capability empowers scientists to observe dynamic cellular processes with outstanding molecular detail. For instance, the team utilized their platform to visualize how cancer cells reorganize their nuclear proteins upon transcriptional inhibition, shedding light on the proteomic rearrangements that underpin cellular response to environmental and pharmacological perturbations.
By generating precise, nanoscale maps of biomolecular distributions in diseased states, this approach offers promising avenues for early disease detection. Subtle alterations in protein interactions and localization, which precede visible symptoms, could serve as biomarkers for cancer and other pathologies, transforming diagnostic paradigms toward earlier and more targeted intervention.
Furthermore, this methodology provides unprecedented insights into the interplay between diverse molecular players within the nucleus. Understanding these intricate relationships is crucial for elucidating mechanisms of gene regulation, chromatin architecture, and the maintenance of genomic stability under physiological and pathological conditions.
The integration of sequence-specific DNA tags with advanced microscopy techniques represents a powerful toolbox for cell biologists and biomedical researchers. It heralds a new era in spatial omics, where the simultaneous imaging of numerous biomolecules at nanoscale resolution paves the way for comprehensive cellular maps, enabling better understanding of complex biological systems in health and disease.
This pioneering study not only enhances the technical capabilities of super-resolution microscopy but also exemplifies the impact of interdisciplinary innovation, where chemistry, molecular biology, and optical physics converge to unravel the complexities of life at the molecular level.
As science continues to push boundaries, technologies like the enhanced DNA-PAINT developed at IISc promise to revolutionize cellular imaging, allowing researchers to peer deeper into the microscopic world and decode the molecular language of cells with unmatched clarity and speed.
Subject of Research: Multiplexed super-resolution imaging of nuclear proteins in cancer cells
Article Title: High-speed multiplexed DNA-PAINT imaging of nuclear organization using an expanded sequence repertoire
News Publication Date: 22-Apr-2026
Web References: http://dx.doi.org/10.1038/s41467-026-72206-0
Image Credits: Mahipal Ganji lab
Keywords: DNA-PAINT, super-resolution microscopy, nuclear organization, biomolecular imaging, transcription machinery, cancer cell imaging, multiplex imaging, nanoscale resolution, fluorescent DNA tags, cellular biomolecules, photodamage reduction, spatial omics
Tags: advanced cellular microscopy methodscancer cell biomolecule visualizationcellular nuclear architectureDNA-PAINT fluorescence microscopygenomic regulation imaginghigh-resolution DNA-PAINT imagingmolecular mapping of nucleusmultiplex biomolecule imagingnanoscale nuclear organizationnuclear structural protein localizationsuper-resolution microscopy techniquestranscription machinery spatial mapping