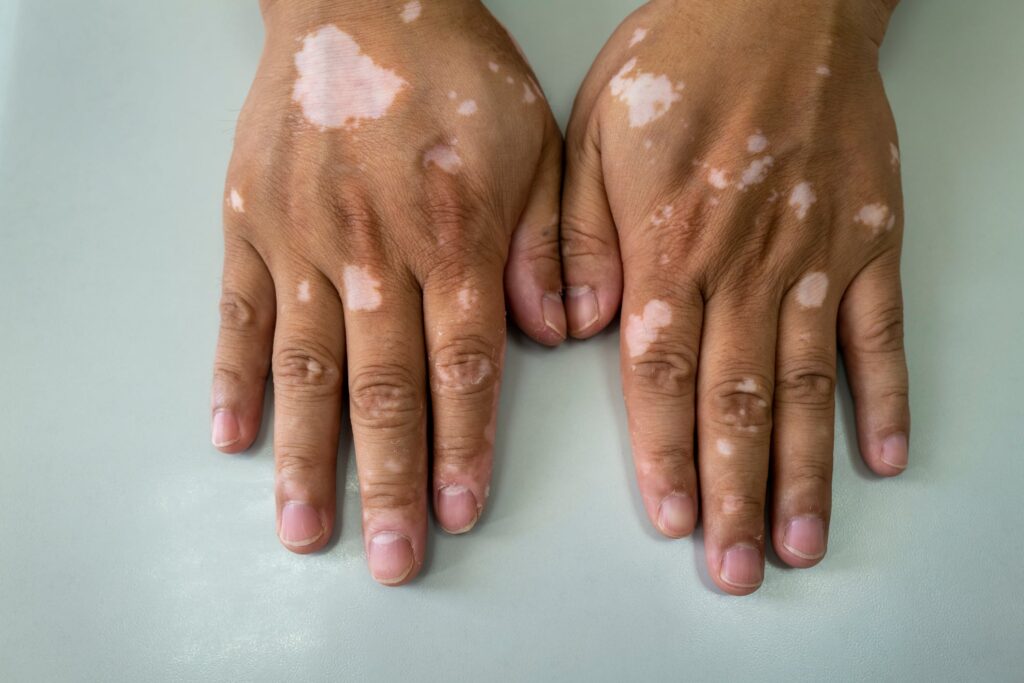
Incyte’s povorcitinib has scored a pair of phase 3 wins for a skin condition, although analysts noted the oral JAK inhibitor didn’t quite live up to AbbVie’s Rinvoq.
Incyte evaluated 30 mg povorcitinib in two late-stage studies, dubbed STOP-V1 and STOP-V2, in adults with nonsegmental vitiligo. Both trials hit their primary endpoint of a more than 75% reduction in a facial vitiligo score at week 52, according to the company’s first-quarter earnings release.
Specifically, 18.9% of povorcitinib-treated patients in both trials hit this 75% mark, compared with 6.8% and 3.1% of patients in STOP-V1 and STOP-V2, respectively.
“Across both studies, statistically significant and clinically meaningful differences were also observed in key secondary endpoint measures,” Incyte explained, including a measure of reducing vitiligo across the body by 50% or more.
Safety and tolerability was consistent with previous studies, with “no new safety signals observed,” the biopharma added.
Incyte will use the latest phase 3 data to support its vitiligo approval push to regulators, which is currently scheduled for the first half of 2027.
In today’s release, Incyte CEO Bill Meury listed the data as one of the signs the company is “building a more durable, growth-oriented portfolio,” alongside hoped-for approvals for povorcitinib to treat another skin condition called hidradenitis suppurativa in Europe and the U.S. in late-2026 and the first quarter of 2027, respectively.
In a note to clients, analysts with William Blair said the “positive” placebo-adjusted benefits of 12% and 16% in the phase 3 trials “should support regulatory filings and approval.”
However, the analysts noted that these findings “will be compared to Rinvoq’s phase 3 data, which showed slightly higher F-VASI75 numbers ranging from 17% to 19% (placebo-adjusted).”
AbbVie read out those results for its blockbuster JAK inhibitor in October 2025. The Big Pharma has since filed regulatory bids with the FDA and the European Medicines Agency with the goal of positioning Rinvoq as potentially the first systemic treatment for the disease.
Related
Vitiligo is a chronic condition where skin loses pigment. Incyte currently markets the only FDA-approved treatment for vitiligo in the form of Opzelura, the topical form of its JAK inhibitor Jakafi.
Aside from vitiligo, Incyte is planning to read out a pair of phase 3 studies for povorcitinib in patients with moderate to severe prurigo nodularis in the fourth quarter of the year, while a phase 2 study in asthma is expected to report topline data in the second half of the year.
Attempts to develop new drugs for the condition ran into trouble last year, with both Incyte and Vyne Therapeutics scrapping candidates.