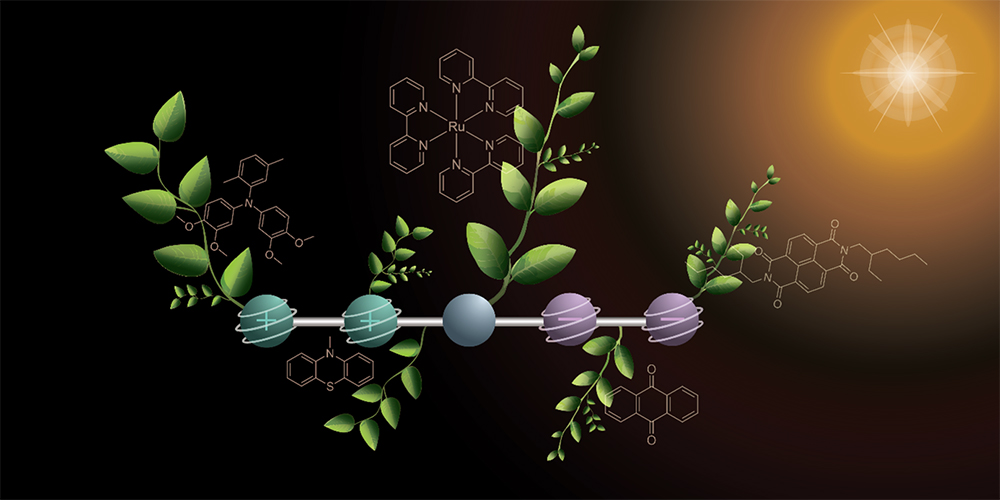
One distinction between natural photochemistry and artificial photosynthesis is the number of electrons transferred at the same time—the latter relies on multi-electron transfer reactions. Now, a group from the University of Basel, Switzerland, has made an important step toward achieving artificial photosynthesis: they developed a molecule that can store four charges—two positive and two negative—simultaneously under light irradiation.
The overarching goal is to convert sunlight into carbon-neutral fuels. However, the new molecule has not yet created a functioning artificial photosynthesis system. “But we have identified and implemented an important piece of the puzzle,” says Oliver Wenger, PhD, professor at the University of Basel. The new findings from the study help to improve the understanding of the electron transfers that are central to artificial photosynthesis. “We hope that this will help us contribute to new prospects for a sustainable energy future,” he adds.
This work is published in Nature Chemistry in the article, “Photoinduced Double Charge Accumulation in a Molecular Compound.”
In addition to being the foundation of virtually all life, photosynthesis could also be the key to environmentally friendly fuels. One area that researchers are exploring is to imitate natural photosynthesis and use sunlight to produce high-energy compounds: solar fuels such as hydrogen, methanol, and synthetic petrol. If burned, they would produce only as much carbon dioxide as was needed to produce the fuels. In other words, they would be carbon-neutral.
The intermediate storage of multiple charges is an important prerequisite for converting sunlight into chemical energy: the charges can be used to drive reactions—for example, to split water into hydrogen and oxygen.
The newly developed molecule reported here—a molecular donor–photosensitizer–acceptor compound in which light absorption leads to the reversible accumulation of two positive and two negative charges—consists of five parts that are linked in a series and each performs a specific task. One side of the molecule has two parts that release electrons and are positively charged in the process. Two on the other side pick up the electrons, which causes them to become negatively charged. In the middle, the chemists placed a component that captures sunlight and starts the electron transfer. More specifically, the authors write that the resulting photoproduct “forms with an overall quantum yield of 37%, has a lifetime of more than 100 ns and stores 3.0 eV of energy.”
In order to generate the four charges, the researchers took a stepwise approach using two flashes of light. The first flash of light hits the molecule and triggers a reaction in which a positive and a negative charge are generated. These charges travel outward to the opposite ends of the molecule. With the second flash of light, the same reaction occurs again, so that the molecule then contains two positive and two negative charges. “This stepwise excitation makes it possible to use significantly dimmer light. As a result, we are already moving close to the intensity of sunlight,” explains Mathis Brändlin, a graduate student in the Wenger lab. “In addition, the charges in the molecule remain stable long enough to be used for further chemical reactions.”
The authors add that this work “represents a step towards more application-oriented research on solar fuels from fundamental studies of photoinduced (single) electron transfer.”