As cell and gene therapies (CGTs) move from the research stage to commercial applications, biomanufacturers are seeing that many of the production techniques they relied upon at lab scale are no longer their best options.
Life sciences solutions providers are well aware of the challenge and are responding with innovations of their own. New solutions include customized reagents that scale from bench to clinical trials, industrialized lentiviral vector manufacturing techniques, and innovations to boost lentiviral transduction.
Scalable reagents

CEO, Teknova
Scalability is one hurdle. “The infrastructure for reagents is set up for blockbuster drugs that are made in 10,000 L batches. But, when you produce CAR T therapeutics and other personalized therapies for rare diseases, you may need only one liter of (a given) reagent…that may have a minimum order of 350 L,” Stephen Gunstream, CEO of Teknova, says. “Generally, these aren’t stock reagents that last a few years. They are made to order [for specific products].”

Manufacturers’ dilemma, therefore, is that the custom, research-grade buffers they used for development lack the Good Manufacturing Practice (GMP)-level safeguards needed for human trials and may not be animal-free, but the animal-free, GMP products developers need aren’t available in small batches.
Continuing to use research-use only (RUO) reagents through scale-up creates other challenges. Aside from the obvious sterility control and consistency concerns, “Changes in mixing and manufacturing during scale-up change the product. Making one liter of product is different from making 1,000 L.” The ability to use the same raw materials and manufacturing processes, but at the appropriate grade, would help manufacturers tremendously.
Flexible small-batch facility
To that point, three years ago, Teknova completed a new facility specifically for small batch, modular manufacturing. It produces custom reagents quickly and at scale and—importantly—at various grades. Using this new facility, “We’ve done custom batches as small as one liter,” Gunstream says.
Step one involved designing a flexible, modular, regulatory-compliant manufacturing facility for RUO, RUO+, and GMP-grade reagents. This enables manufacturers to use the same reagents start to finish, selecting the appropriate grade for each step. That ability minimizes reagent costs, reduces risks, increases speed to market, and enhances product quality and consistency.
The facility, Gunstream points out, has been validated under multiple production scenarios, and features a “robust microbial contamination strategy that scales across manufacturing grades.”
Things you didn’t know you’d make
Teknova employs a bracketed validation strategy to create “a quality system designed to work with things you didn’t know you’d be making,” Gunstream says. That entailed analyzing raw materials used for custom reagents, knowing which have been used successfully in approved products, and validating these materials themselves at certain ranges.
To smooth development and scale-up, Gunstream advises selecting a supplier that:
- Can scale from beginning to clinical trials
- Is flexible and fast
- Has experience making small, custom batches
- Shares their insights
Manufacturing LVVs at scale
With roughly one dozen CAR T therapies in or nearing clinical trials, no single delivery technology dominates.
“From a manufacturing standpoint, this [plethora of options] places strong demands on vector production, purification, analytics, and scalability. As the field evolves, robust, flexible manufacturing platforms are essential,” Brian Tomkowicz, PhD, vice president and head of R&D and virology fellow, SK pharmteco, said in a recent webinar.
SK pharmteco is optimizing production of lentiviral vectors (LVVs), one of the go-to delivery CGT options, by industrializing ex vivo LVV production even as CAR T therapeutic delivery shifts toward in vivo modalities. With that shift, LVVs are defined as a drug product rather than an intermediate. Consequently, “The demands placed on antiviral vector manufacturing are shifting dramatically,” Tomkowicz said.
“An under-appreciated reality in LVV manufacturing is that downstream yield is overwhelmingly constrained by ion exchange chromatography (IEX) recovery,” he pointed out. “Although upstream harvest and clarification stems preserve the majority of vector material, a substantial loss occurs during the ion exchange step…even in processes that are optimized.” Consequently, “Total [functional particle] recovery often plateaus around 15% to 40%, with 20% being typical.”
In contrast, SK pharmateco’s high-throughput convective IEX delivers a functional particle recovery rate that ranges from 60% to 90%. Charts show a 98% host cell protein clearance and a transducing unit recovery rate exceeding 85% across multiple runs. To obtain such results, SK pharmateco replaced IEX’s traditional packed-bed resin with a membrane adsorber.
IEX, using a membrane absorber, cut total processing time to one hour or less (down from two to four hours using resin). That’s thanks in part to load capacities up to 250 mL/MV and residence times of less than six seconds, which help make throughput more than five times faster than packed-bed resin-based IEX. “This enables same-day processing,” Tomkowicz said.
Importantly, this high-throughput, membrane absorber-based IEX performance is maintained as process stress increases. Multiple tests involving a variety of input concentrations and higher viral particles measured physical recovery and functional titers. The highest levels of recovery peaked at 60–85%.
“Membrane [IEX] enables smaller footprints, disposable formats, and substantially lower material and buffer costs across both the 50L and 500 L production scales. This results in a six- to 20-fold cost reduction,” Tomkowicz explained.
The total cost of producing a 50 L batch using membrane adsorber IEX is about $3,000, he says, down from the $60,000 to $70,000 cost of a resin-based approach.
The most important optimization lesson, he said, is that “Recovery is governed by mass transfer physics rather than brand-specific attributes.”
As the industry moves from ex vivo to in vivo modalities, “The key difference is not just how we manufacture an LVV, but what we package inside it. Therefore, architecture and rational payload design become essential to allow manufacturers to exploit variants,” Tomkowicz emphasized. “While our focus is on scalable manufacturing, payload design and process requests must develop together.”
Tomkowicz called this a simple, scalable, fit-for-purpose platform for clinical and commercial manufacturing, predicting it will be “a manufacturing inflection point.” Before this method is widely adopted, however, Tomkowicz said he would like to see the work expanded to more vectors and more patients.
LVVs for adoptive cell therapy

LentiBOOST business leader
Revvity
“Perhaps the greatest challenge in the field of CGT today is the high cost of developing and manufacturing lentiviral vectors,” Chris Lowe, PhD, LentiBOOST business leader at Revvity, says. “One of the key drivers of this cost is the difficulty in achieving reproducible lentiviral transduction efficiency across a range of clinically relevant cell types (including T cell, hematopoietic stem cells, and progenitor cells) without compromising viability, phenotype, or function,” he tells GEN.
The issue manufacturers face is that even optimized vectors may require a high copy number if transduction is inefficient. “Transduction efficiency, therefore, can be the difference between a program that reaches the clinic and one that gets stuck in the lab because vector supply, manufacturing costs, or product quality are not sustainable at scale,” Lowe points out.
To address this challenge, industry scientists are exploring approaches that include optimizing vector design and production, improving physical and process-based activities, and developing chemical and biological enhancers.
For example, transduction can be improved by refining envelope pseudotypes, promoter selection, and genome architecture to improve tropism, especially when those technologies are accompanied by advances in vector manufacturing that increase functional titer and consistency. “While beneficial,” Lowe says, “these strategies enhance transduction indirectly and can increase manufacturing complexity.”
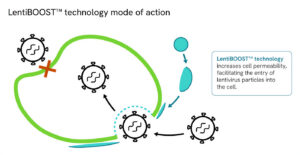
Likewise, spinoculation, controlled temperature shifts, and other physical and process-based methods to increase virus-cell contact (often combined with media and serum optimization) may improve cell fitness during transduction but are not without their own challenges. “Specifically,” Lowe cautions, “care must be taken to avoid issues with cell health, and these techniques can be challenging to scale up. These methods are often best paired with complementary chemical and biological enhancers.”
Such enhancers typically increase lentiviral transduction by promoting virus-cell interaction of membrane fusion, Lowe elaborates. “Traditional approaches such as polycations and extracellular matrix components are well established but can be associated with cytotoxicity, activation of cellular stress pathways, variable performance across primary cell types, and limited availability in consistent GMP-grade formats.
“Polymer-based enhancers offer an alternative approach that addresses some of these limitations,” Lowe continues. “Non-ionic poloxamer formulations, including technologies such as [Revvity’s] LentiBOOST™ enhancer, have been shown to improve transduction efficiency and increase vector copy number while maintaining cell viability.”
By providing a receptor-independent way to modulate the interface between viral and cellular membranes, the poloxamer in Revvity’s LentiBOOST technology promotes fusion. This increases the odds that each particle will deliver its cargo successfully across lentiviral constructs and multiple cell types—including primary T cells and human stem cells.
Multiple options
Today the industry is exploring “a variety of different approaches to enhance lentiviral transduction, including nanoparticle-assisted delivery, transient pathway modulation, and next-generation surface or matrix technologies that improve virus—cell contact or intracellular trafficking,” Lowe says.
That said, there are strong reasons to optimize the cellular environment. “Lentiviral transduction is most efficient when cells are metabolically active, not stressed, and can support membrane fusion and reverse transcription,” he says. Revvity, through BioLegend, which it acquired in 2021, also offers cell culture reagents in its Cell Vive™ family.
As CGT developers look to increasingly challenging cell types and ever-more-complex engineering, “there is a corresponding need to improve transduction efficiency…to increase therapeutic response rates, and to lower manufacturing costs,” Lowe says.
In the future, he adds, “Lentiviral transduction will increasingly coexist with non-viral and hybrid delivery systems. In that context, effective, GMP-grade enhancers will help ensure that lentiviral vectors remain a cornerstone technology where stable, durable gene delivery is required.”
With the plethora of advances being developed today, CGT manufacturers are on the cusp of improvements that will reduce process times, cut costs, enhance flexibility, and support scalability.
Fouad Atouf, PhD, is the chief science officer at United States Pharmacopeia.
References
1. Murimi-Worstell B, Ballreich M, Seamans G, Alexander, C. Association between US Pharmacopeia (USP) monograph standards, generic entry and prescription drug costs. Published: November 12, 2019.
2. Gupta, RK. The vital role of biological standardization in ensuring efficacy and safety of biological products–Historical perspectives. Journal of Pharmaceutical Sciences, 2025; 114(2): 690–700.
3. Atouf F and Venema J. Do Standards Matter? What is Their Value? Journal of Pharmaceutical Sciences, 2020; 109(8): 2387-2392.