Sponsored content brought to you by
Research approaches are becoming increasingly complex with the rise of multiomic methods and artificial intelligence (AI) integration. In fact, today’s experimental designs and research technologies are so advanced that they are beginning to match the complexity of the biological systems and diseases that researchers are trying to understand.
While some diseases are well characterized, others display significant heterogeneity at molecular and cellular levels. Cancer is perhaps the clearest example, differing widely between individuals even when well-characterized genomic features are shared. Researchers have long recognized that a one-size-fits-all approach cannot be applied to cancer. However, despite significant advances in identifying and characterizing cancer subtypes, treatment personalization remains largely limited. And the tools and infrastructure needed to accurately dissect tumor complexity have historically been lacking.
The Many Layers of Tumor Diversity
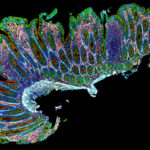
Tumor heterogeneity is influenced by factors such as the cell type from which the tumor develops, the tissue of origin, and the degree of infiltration by other cell types, including immune cells. Intratumoral location also plays an important role, as cells can behave differently depending on local conditions such as oxygen and nutrient gradients. These differences are further compounded as tumors evolve and respond to treatment. The importance of gaining a better understanding of cancer heterogeneity is highlighted both by the limited responses of many patients to targeted therapies and by a growing appreciation of the roles different cell types play in tumor progression. Without the tools to understand critical differences, it remains difficult to predict which patients will benefit from specific treatments and to identify new avenues for treatment development. This picture is changing, however, with the growing availability and adoption of spatial biology approaches.
Incorporating Spatial Insights into Oncology Research
Spatial biology is becoming a key technique for dissecting the cellular makeup and tissue architecture of tumors. It allows researchers to identify not only which cells are present and where they are located but also gain insight into their function. By capturing the diversity of tumor-resident cell types and their spatial relationships, spatial biology generates rich datasets with important implications for therapeutic strategy, biomarker development, and patient stratification. Incorporating spatial context is thus essential for translating complex tumor biology into targeted and immune-based therapies.
Tumors constantly adapt to pressures such as immune attack and different therapies. Thus, there is immense value in capturing spatial tumor heterogeneity at different stages, particularly pre- and post-treatment in clinical trials. In a recent paper published in Cell,1 researchers used spatial biology approaches to show that oncolytic virus treatment in glioblastoma can drive spatially localized T cell-mediated tumor killing associated with improved patient survival in a clinical trial, highlighting how spatial insights reveal interactions between immune and tumor cells that influence patient outcomes.
Several challenges remain. Large cohort studies require a robust and scalable logistical pathway spanning sample collection, processing, storage, and analysis. When these steps are fragmented across multiple vendors, it can lead to delays, higher costs, and the introduction of preanalytical variation that confound results.
Reducing Variability, Improving Reproducibility
Spatial biology holds unique promise for improving our understanding of cancer and other complex diseases. But applying these technologies successfully in oncology clinical trials requires a strong operational foundation for standardized sample collection, timely processing, high-quality analysis, and long-term storage. As a CAP and CLIA accredited integrated laboratory and biorepository, Sampled provides end-to-end support for sample collection and analysis through storage, within a single coordinated environment. Equipped with the full 10x Genomics suite in-house, including the Chromium, Visium, and Xenium instruments, Sampled empowers researchers to apply spatial biology at scale and generate more robust datasets for oncology studies.
Reference
1. Meylan M, Tian Y, Wu L, et al. Persistent T Cell Activation and Cytotoxicity against Glioblastoma Following Single Oncolytic Virus Treatment in a Clinical Trial. Cell 2026

Visit sampled.com/gen.