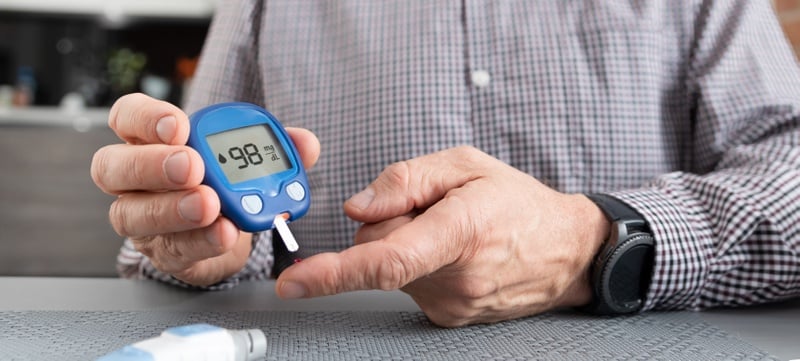
The FDA is flagging a high-risk issue with blood glucose monitors manufactured by Trividia Health that the company says is tied to 114 serious injuries and one death since the devices’ launch 12 years ago.
The agency’s communication comes after Trividia sent an urgent letter (PDF) to customers back on Feb. 6 updating the owner’s booklet and instructions for the affected systems.
The device measures blood glucose through a fingerprick blood sample placed on a test strip. According to the letter, the devices may display an E-5 error code for a very high blood glucose event (> 600 mg/dL) or when there is a test strip error.
As originally written, the instructions for the error code could lead to a potentially dangerous delay in treatment that “may result in serious adverse health consequences, such as dehydration, altered mental status, or death, especially for users with very high blood glucose levels,” according to the letter.
The updated instructions emphasize that users should seek medical attention immediately if they receive the error code and are experiencing symptoms of high glucose such as fatigue, excessive urination, thirst or blurry vision, the company said.
The alert includes (PDF) all True Metrix, True Metrix Air, True Metrix Go and True Metrix Pro devices sold in the U.S., the U.K., Mexico, Australia and the Caribbean. It also includes store-branded versions of the device sold at pharmacies including CVS, Rite Aid and Walgreens.
Trividia said devices do not need to be returned, and customers may continue to use them.
The FDA’s early alert comes as part of a new initiative that aims to provide speedier communication to the public about potentially high-risk medical device issues. The agency said it is continuing to review information about the issue and will update its alert if new information becomes available.