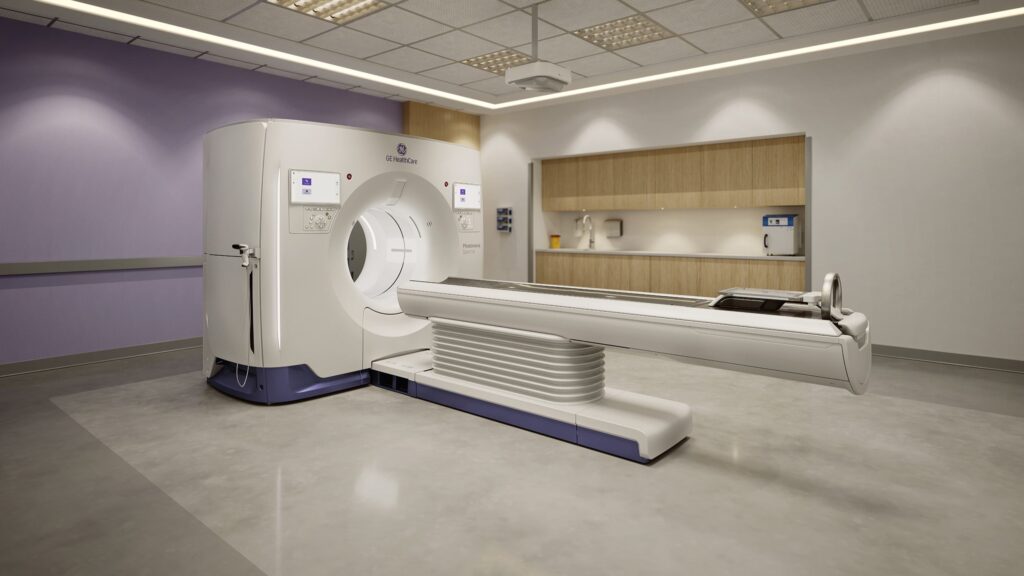
Following its curtain-raising at last fall’s meeting of the Radiological Society of North America, GE HealthCare has obtained an FDA clearance for its photon-counting CT system.
The new Photonova Spectra aims to capture up to 50 times more data than some of its premium CT predecessors. The company said the scanner is the product of a $5.1 billion investment in R&D spanning decades, including the 2020 acquisition of the developer of its detector technology, dubbed Deep Silicon.
GE HealthCare said it has tapped Photonova Spectra to spearhead a new generation of products within its imaging portfolio, which it expects will eventually add 1% to 2% in revenue growth.
Compared to conventional CT, which converts X-ray beams into visible light before evaluating them, photon-counting machines directly measure their energy after they pass through the body, allowing higher resolution and clearer images of different types of tissue.
Related
Deep Silicon detectors, initially developed by the Swedish startup Prismatic Sensors, aim to eliminate excess electronic noise during a scan while reducing the overall radiation dose delivered to the patient. The system is also supported by Nvidia GPUs for image processing.
Paired with gantry hardware that can rotate around the body in 0.23 seconds, GE HealthCare said that Photonova Spectra can help distinguish between materials such as calcium, fat and iodine, the latter a key injection to boost X-ray contrast.
The company said the system can be used across medical specialties—such as neurology and oncology, as well as in musculoskeletal, thoracic and cardiac exams—while supporting a one-scan approach that reduces the number of protocols and setup time.
The University of Wisconsin-Madison has served as the first U.S. clinical evaluation site for the scanner, while Stanford Medicine has contributed to human subject and technical research programs.
Related
“Elevating diagnostic confidence, particularly with subtle low-contrast structures, requires increasing clarity significantly to facilitate enhanced material differentiation,” Giuseppe Toia, M.D., CT modality chief at the University of Wisconsin School of Medicine and Public Health, said in a statement.
“Being involved in developing and testing the Deep Silicon detector has allowed us to see what this technology is capable of. During evaluative studies, this system allowed us to extract more clinically actionable information from a single scan to support informed decisions for research and clinical care,” Toia added. “Because photon counting CT is a fundamentally different approach to imaging, we find it results in clean spectral signatures, high spatial resolution and accurate CT numbers.”