A study led by Adelaide University scientists at the Centre for Cancer Biology (CCB) has uncovered a critical mechanism driving the growth and spread of glioblastoma (GBM)—one of the most aggressive and lethal forms of brain cancer—potentially paving the way for more effective treatments.
Studying human tissues and mouse models, the team identified a previously unrecognized role for CD47 in promoting tumor progression, independent of its known function in helping cancer evade the immune system. The new research, published recently, shows that CD47, already a target of experimental cancer immunotherapies, also plays a direct role inside tumor cells, helping them grow, move, and invade surrounding brain tissue. The findings highlight a signaling pathway that may represent a potential therapeutic target for glioblastoma.
“We’ve known for some time that CD47 acts as a kind of ‘don’t eat me’ signal that helps cancer cells hide from the immune system,” said research co-lead Nirmal Robinson, PhD. “What we’ve discovered is that CD47 is doing much more than that; it’s actually driving the cancer’s ability to spread and grow.”
Senior author Robinson and colleagues described their findings in PNAS, in a paper titled “CD47 stabilizes ROBO2 to regulate glioblastoma progression by preventing ITCH-mediated ubiquitination,” in which they concluded “… these findings reposition CD47 as a driver of GBM plasticity and highlight the CD47–ROBO2 axis as a therapeutic target.”
Glioblastoma is the most common and aggressive form of primary brain tumor, and is extremely difficult to treat, with most patients surviving less than 18 months after diagnosis. Current treatments—including surgery, radiation and chemotherapy—offer only limited benefit, and the cancer almost always returns. “Therefore, there is an urgent need for novel treatment strategies that have greater specificity and efficacy,” the authors wrote.
CD47, a ubiquitously expressed cell surface receptor, functions as a critical innate immune checkpoint regulator, and is found to be overexpressed in many cancers, including GBM. “CD47 is an innate immune checkpoint that inhibits phagocytosis by myeloid cells, contributing to immune evasion by cancer cells,” the team explained. Robinson added, “We’ve known for some time that CD47 acts as a kind of ‘don’t eat me’ signal that helps cancer cells hide from the immune system.”
Recent studies have demonstrated that CD47 has cell-intrinsic functions beyond its role in promoting immune evasion, and suggest that CD47 exerts immune-independent effects to facilitate cancer progression. “In this study, we investigated the cell-intrinsic role of CD47 in driving GBM progression,” the researchers said.
Through their newly reported research the team initially found that CD47 is particularly abundant at the invasive edges of glioblastoma tumors—the regions responsible for cancer spread into healthy brain tissue. Patient data also showed that those with higher levels of CD47 had significantly poorer survival outcomes.
In collaboration with Stuart Pitson, PhD’s CCB team, the researchers then used a combination of laboratory experiments and animal models to show that removing or blocking CD47 dramatically reduced tumor cell proliferation, migration and invasion. Tumors lacking CD47 grew more slowly and, in some cases, survival time in animal models nearly doubled. “We demonstrate that CD47 loss impairs GBM cell proliferation, migration, and invasion, independent of immune activity, and leads to reduced tumor burden and prolonged survival in vivo,” the team stated.
These effects occurred even in the absence of immune cells, confirming that CD47 has a powerful tumor-promoting role beyond immune evasion. The team in addition identified a key partner protein, ROBO2, which acts downstream of CD47 and contributes to glioblastoma growth and spread. “Functionally, CD47 drives GBM cell proliferation and migration through activation of the ROBO2 signaling pathway,” they added. “Loss of CD47 or ROBO2 in GBM cells led to significantly reduced tumor growth and prolonged survival in intracranial xenograft mouse models.”
The researchers discovered that CD47 protects ROBO2 from being broken down inside the cell. It does this by sequestering a protein called ITCH, which would otherwise tag ROBO2 for destruction. “Mechanistically, we demonstrate that CD47 stabilizes ROBO2 protein levels by preventing its degradation. Specifically, CD47 sequesters the E3 ubiquitin ligase ITCH, thereby inhibiting ITCH-mediated proteasomal degradation of ROBO2.”
Ruhi Polara, PhD, who led the research alongside Robinson, further commented, “Essentially, CD47 is shielding ROBO2, allowing it to accumulate and drive tumor progression. When we remove CD47, ROBO2 is degraded, and the cancer cells lose their ability to grow and invade effectively.”
The findings reveal a previously unknown molecular pathway—CD47–ITCH–ROBO2—that controls how glioblastoma cells behave. This opens up new possibilities for treatment strategies that go beyond current approaches. While therapies targeting CD47 are already being tested in clinical trials for other cancers, they have shown limited success in glioblastoma so far. The new research suggests that directly targeting the CD47–ROBO2 pathway, or disrupting the stabilisation of ROBO2, could be a more effective strategy. “In summary, our study reveals a role of CD47 in regulating cellular plasticity suggesting that targeting ROBO2 could offer a promising alternative therapeutic strategy for GBM,” they stated.
“By understanding this mechanism, we now have new targets to explore,” Polara said. “This could lead to the development of therapies that specifically block the tumor’s ability to spread, which is one of the biggest challenges in treating glioblastoma.”
The study also highlights the importance of looking beyond the immune system when developing cancer treatments. “This work changes how we think about CD47,” Robinson said. “It’s not just an immune checkpoint; it’s a central regulator of tumor biology in its own right.”
The researchers say further studies are needed to translate these findings into clinical treatments, but the discovery represents a significant step forward in the fight against one of the most devastating cancers.

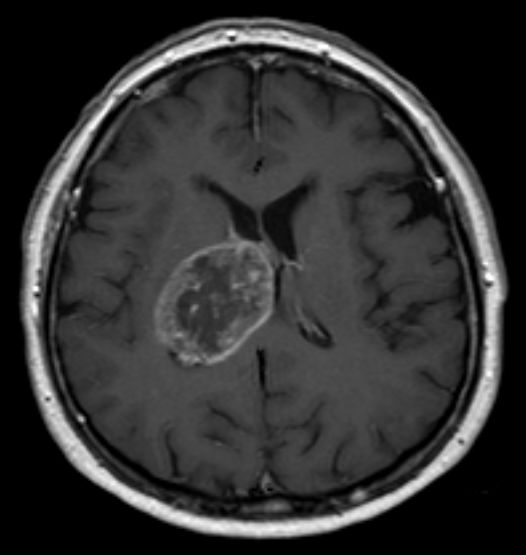
![Glioblastoma [Wikimedia Commons] Glioblastoma [Wikimedia Commons]](https://www.genengnews.com/wp-content/uploads/2018/08/Glioblastoma_multiforme__MRT_T1KM_ax2212041010.jpg)