In a groundbreaking study set to reshape our understanding of obesity-related inflammation, researchers have unveiled the pivotal role of a collagen-specific molecular chaperone called Heat Shock Protein 47 (Hsp47) within inguinal white adipose tissue (iWAT). This protein has been shown to amplify inflammatory gene expression in response to a high-fat diet, specifically in male mice, shedding new light on the molecular intricacies governing metabolic inflammation and its pathological consequences. The findings promise to open novel avenues for targeted therapies against obesity-related diseases, which remain among the most pressing global health challenges today.
The molecular chaperone Hsp47 is known for its critical function in collagen biosynthesis and ensuring proper folding and assembly of collagen triple helices. Collagen, a structural protein that is abundant in the extracellular matrix (ECM), plays an essential role in maintaining tissue architecture and function. While the role of ECM remodeling has been previously implicated in adipose tissue dysfunction, the specific involvement of Hsp47 within adipose depots had remained elusive until this recent investigation illuminated its contribution to metabolic inflammation.
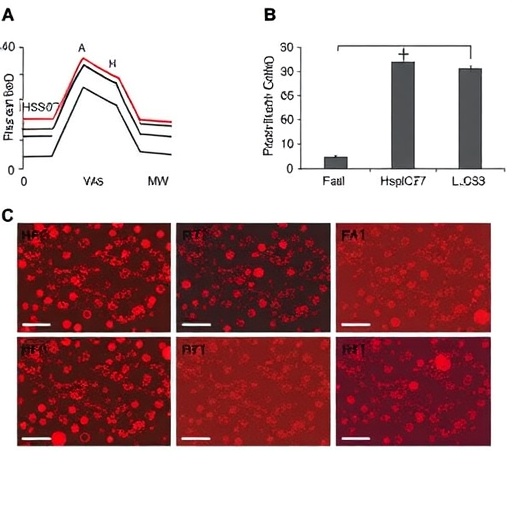
Inguinal white adipose tissue, positioned subcutaneously, serves as an important energy reservoir and an endocrine organ, regulating systemic metabolism through secreted adipokines and cytokines. The study meticulously focused on this depot, uncovering that Hsp47 expression escalates significantly in male mice subjected to prolonged high-fat diet feeding. This upregulation corresponded with an increase in inflammatory gene expression, signaling that Hsp47 may act as a crucial molecular switch in the adipose tissue inflammatory milieu under conditions of nutrient excess.
Remarkably, the research delineated the mechanistic pathway by which Hsp47 influences inflammation, highlighting its role in modulating ECM dynamics and cellular signaling pathways related to fibrosis and immune cell recruitment. The overexpression of Hsp47 promotes collagen deposition, which structurally remodels the iWAT microenvironment — a change that invariably fosters pro-inflammatory gene expression. This cascade underscores how ECM alterations extend beyond mere scaffolding to actively participate in the tissue’s immunometabolic responses.
The study further detailed that the interaction between Hsp47-mediated collagen accumulation and inflammatory signaling likely involves crosstalk with resident macrophages and other immune components in the adipose tissue. Macrophages, known to populate expanding adipose tissues during obesity, adopt a pro-inflammatory phenotype contributing to chronic low-grade inflammation, insulin resistance, and metabolic dysfunction. The findings suggest that Hsp47-driven ECM shifts create a permissive niche for these immune cells to perpetuate inflammation.
Importantly, sex-specific differences emerged from this research, with male mice displaying a more pronounced relationship between Hsp47 expression and inflammation in response to a high-fat diet. This intriguing sex dimorphism demands further exploration, as it may offer insights into gender-based susceptibilities in metabolic diseases and guide personalized intervention strategies.
From a translational medicine perspective, these discoveries hold tremendous promise. The specific targeting of Hsp47 or its downstream signaling pathways could attenuate pathological collagen remodeling and disrupt the vicious cycle of inflammation and fibrosis in adipose tissue. This strategy offers a potential to halt or reverse metabolic derangements before clinical manifestations such as type 2 diabetes or cardiovascular diseases ensue.
Moreover, the study employed advanced molecular biology techniques, including gene expression profiling, immunohistochemistry, and protein interaction assays, to comprehensively characterize the expression patterns and biological functions of Hsp47 in adipose tissue. These robust methods ensured that the conclusions drawn are grounded in a thorough experimental framework, enhancing their credibility and potential for future validation in clinical contexts.
The implications of this research extend beyond obesity alone. Since collagen remodeling and inflammatory processes are fundamental to a variety of pathological states, including fibrosis in other organs and cancer progression, understanding the role of Hsp47 could illuminate shared molecular pathways among these conditions. This places Hsp47 not only as a central figure in adipose tissue biology but also as a broader biomolecular target with wide-reaching clinical implications.
Furthermore, these findings encourage a paradigm shift in adipose tissue research — emphasizing the ECM’s dynamic role as an active participant rather than a passive structure in metabolic regulation. This conceptual change could inspire innovative experimental designs focusing on the triad of cellular, molecular, and extracellular interactions within metabolic tissues.
The researchers also hypothesize that dietary modulation could influence Hsp47 expression, suggesting that lifestyle interventions might mitigate ECM-related inflammation in adipose tissue. This link underscores the complex interplay between nutrition, molecular chaperones, and tissue health, which could be exploited for more effective public health strategies combating obesity and its complications.
Interestingly, the work aligns with emerging evidence on the role of molecular chaperones in stress responses and their contribution to maintaining cellular proteostasis under metabolic stress. Hsp47’s collagen-specific role adds a new dimension to this narrative by connecting molecular chaperoning directly to tissue structure and inflammatory regulation under high-fat dietary conditions.
Finally, this study paves the way for further investigations into pharmacological agents or genetic tools aimed at modulating Hsp47 activity. Such future endeavors could explore whether Hsp47 inhibition ameliorates inflammation and metabolic dysfunction in vivo, potentially culminating in novel therapeutic options that address both obesity’s molecular causes and its inflammatory sequelae.
In conclusion, the elucidation of Hsp47’s role within inguinal white adipose tissue enriches our understanding of how extracellular matrix remodeling intersects with inflammatory gene activation during high-fat diet challenges. This discovery positions Hsp47 as a promising molecular target to disrupt harmful adipose tissue remodeling and chronic inflammation, with hopes to curb the global burden of obesity and metabolic diseases. As the scientific community continues to decipher adipose tissue’s complex biology, such insights reinforce the importance of integrating molecular chaperones and ECM dynamics into the metabolic disease narrative, fostering innovative therapeutic quests on a path toward healthier futures.
Subject of Research: Collagen-specific molecular chaperone Hsp47 in inguinal white adipose tissue and its role in high-fat diet-induced inflammatory gene expression.
Article Title: Collagen-specific molecular chaperone Hsp47 in inguinal white adipose tissue promotes high-fat diet-induced inflammatory gene expression in male mice.
Article References:
Ito, S., Kamei, R., Kasai, A. et al. Collagen-specific molecular chaperone Hsp47 in inguinal white adipose tissue promotes high-fat diet-induced inflammatory gene expression in male mice. Sci Rep (2026). https://doi.org/10.1038/s41598-026-45003-4
Image Credits: AI Generated
Tags: adipokines and cytokines in fat tissueadipose tissue extracellular matrix in metabolic diseasecollagen biosynthesis and ECM remodelingcollagen molecular chaperone in obesitydiet-induced inflammation mechanismshigh-fat diet effects on iWATHsp47 role in adipose tissue inflammationinguinal white adipose tissue functionmetabolic inflammation in obesitymolecular targets for obesity-related diseasessex-specific inflammation in obesitytherapeutic strategies for metabolic inflammation