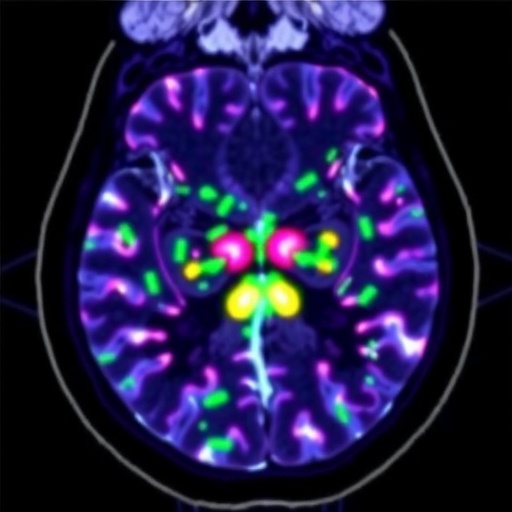
A groundbreaking study from King’s College London has unveiled an innovative imaging tracer that uniquely illuminates the role of fats in tumor metabolism, advancing our understanding of cancer growth and offering promising avenues for therapeutic development. This novel tracer sheds light on the metabolic preferences of tumors, particularly their surprising reliance on fatty acids as an energy source, a mechanism previously overshadowed by the well-known glucose consumption patterns of cancer cells.
Living organisms depend on a dynamic array of nutrients to sustain cellular energy demands, with carbohydrates, fats, and proteins each playing critical roles under varying physiological conditions. However, in pathological states such as cancer and cardiovascular disease, these metabolic pathways undergo significant shifts. Traditionally, research and clinical imaging have focused primarily on glucose metabolism using tracers like fluorodeoxyglucose (FDG), given the high glycolytic activity of many tumors. Yet, this new approach pioneers the visualization of lipid metabolism in vivo, a frontier that has remained relatively elusive until now.
The research team, helmed by Professor Tim Witney from the School of Biomedical Engineering & Imaging Sciences, engineered a tracer based on carnitine, a pivotal molecule in fatty acid metabolism. Carnitine facilitates the shuttling of long-chain fatty acids across the mitochondrial membrane, a critical step for β-oxidation and subsequent ATP production within the cell. By tagging carnitine with a radioactive isotope, the team created a unique PET imaging agent capable of revealing real-time fatty acid utilization in living organisms, thereby providing an unprecedented window into metabolic fluxes associated with health and disease.
This advancement was rigorously demonstrated through preclinical models that showcased differential uptake of the carnitine-based tracer in various tissues, confirming its capacity to map lipid metabolism in physiological and pathological contexts. In particular, the tracer’s uptake was markedly elevated in certain aggressive tumor subtypes, emphasizing these cancers’ ability to harness fatty acids for energy production alongside glucose. Such findings challenge the entrenched view that cancer cells predominantly rely on glycolysis, opening new perspectives on tumor biology and metabolic heterogeneity.
One significant implication of this tracer lies in its potential to refine cancer diagnostics and treatment monitoring. Whereas current metabolic imaging predominantly captures glucose utilization, this dual insight into lipid metabolism may identify tumors that exploit alternative fuel sources, which could be inherently resistant to therapies targeting glycolysis. Consequently, this technology could assist in stratifying patients more effectively and tailoring metabolic interventions to disrupt cancer growth more efficiently.
Beyond oncology, the team’s findings elucidate important metabolic alterations across cardiovascular diseases where carnitine homeostasis is often disrupted. Shifts in carnitine metabolism observed in cardiac tissues may serve as early biomarkers for disease progression, enabling interventions at stages when clinical symptoms have yet to manifest fully. Such capability could revolutionize patient outcomes by facilitating timely therapeutic responses grounded in metabolic evidence.
Furthermore, this carnitine-based tracer carries broad prospects extending into sports science and metabolic health research. By visualizing fatty acid metabolism with precision, researchers can assess how physical training or nutritional supplementation affects energy substrate selection and mitochondrial function. The tracer offers the potential to quantify the metabolic impact of carnitine supplements, widely popular for purported benefits in enhancing athletic endurance and recovery.
The study’s success underscores the intersection of molecular imaging, biochemistry, and clinical research, embodying an integrative approach to uncovering fundamental biological processes. It highlights the role of sophisticated engineering in designing molecular probes that not only trace cellular activities but also inform therapeutic strategies tailored to metabolic nuances across diseases.
Featuring as the cover article in the prestigious journal Advanced Science, this work accentuates the innovative capabilities of the Witney Lab’s research, reflecting years of painstaking experimentation led by co-first authors Dr. Richard Edwards and Dr. Ella-May Hards. Their collaboration exemplifies the pioneering spirit required to translate molecular discoveries into clinically actionable imaging tools.
This cutting-edge tracer also serves as a testament to the versatility of carnitine as a biochemical sentinel, capable of capturing the metabolic dialogue between mitochondrial function and nutrient availability. By extending imaging beyond glucose-centric paradigms, the tracer propels forward the frontiers of personalized medicine and metabolic diagnostics, positioning itself as a versatile platform for ongoing and future investigations into cellular energetics.
In practical terms, the integration of this tracer into clinical workflows could transform the management of complex diseases characterized by metabolic dysregulation. From anticipating tumor behavior to monitoring cardiac metabolic health, the tracer offers a powerful lens into biological function that current modalities cannot match, promising a new era of metabolic precision medicine.
In summary, this King’s College London study represents a monumental leap in metabolic imaging technology. By harnessing radiolabeled carnitine, it delivers unprecedented insights into fatty acid metabolism in vivo, revealing critical metabolic reprogramming in cancer and cardiac pathologies. It stands poised to revolutionize diagnostic imaging and therapeutic targeting, underscoring the vital role fats play in health and disease alongside the classic emphasis on glucose.
Subject of Research: Development and application of a novel carnitine-based imaging tracer to visualize fatty acid metabolism in tumors and cardiac diseases.
Article Title: Visualizing Fatty Acid Metabolism in Cancer and Cardiac Disease Using a Radiolabeled Carnitine Tracer
Web References: https://advanced.onlinelibrary.wiley.com/doi/10.1002/advs.202514668
Keywords: cancer metabolism, fatty acid metabolism, carnitine, molecular imaging, PET tracer, metabolic reprogramming, tumor energetics, cardiac metabolism, precision medicine, radiotracer development, mitochondrial function, metabolic diagnostics
Tags: advances in tumor imaging technologiesbiomedical imaging sciences researchcancer metabolic pathwayscarnitine-based imaging tracerfatty acid energy source in tumorsfatty acid metabolism in tumorsinnovative cancer imaging tracerKing’s College London cancer studylipid metabolism visualization in vivometabolic preferences of cancer cellstherapeutic development for cancer metabolismtumor fat-fueled growth mechanism