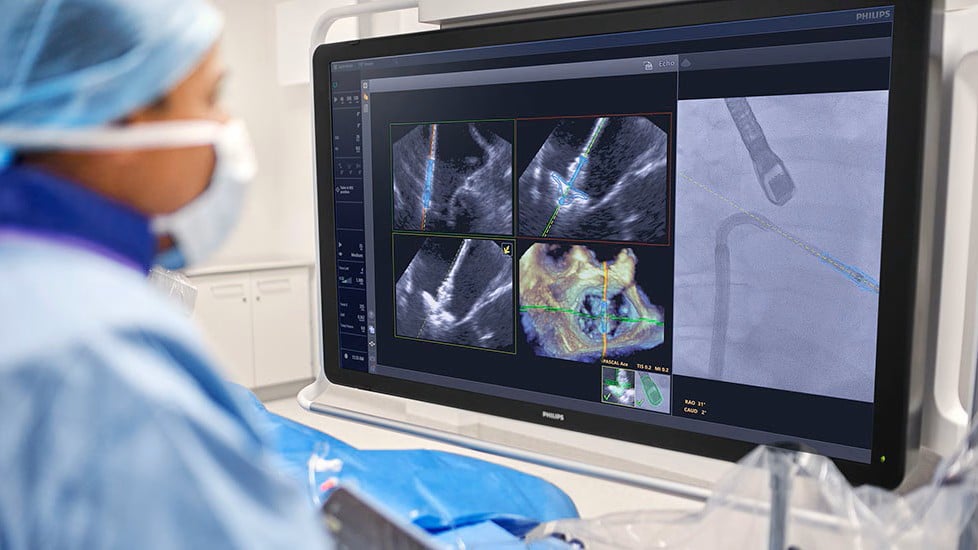
Philips has secured an FDA clearance for its artificial intelligence program that tracks and guides the placement of a minimally invasive heart valve implant in real time.
DeviceGuide is aimed at one of the cath lab’s more challenging procedures, transcatheter mitral valve repair, where a clip-like implant is threaded into a beating heart’s leaking mitral valve to grasp its leaflets and deliver a tighter and more efficient seal.
Developed in collaboration with Edwards Lifesciences, the AI program pairs with the valve maker’s Pascal Ace transcatheter repair implant for mitral regurgitation. DeviceGuide works with Philips’ Azurion image-guided therapy system and EchoNavigator platform, which combines ultrasound and X-ray scans to build a 3D model of the patient’s heart and automatically visualizes the trajectory and placement of the clip.
“The AI software serves as an assistive tool; the physician always remains in control. This isn’t about replacing expertise—it’s about amplifying it,” Atul Gupta, M.D., Philips’ chief medical officer for diagnosis and treatment, said in a statement. “By embedding AI into the procedure, DeviceGuide gives physicians an extra pair of eyes, helping them treat more patients safely and confidently.”
Related
The curtain was raised on DeviceGuide last November, during the London Valves annual structural heart meeting. Now, with the FDA’s blessing, Philips said it plans to showcase the tech at the American College of Cardiology conference this weekend in New Orleans.
According to a case report published last December in JACC, having interventional cardiologists manually track the movement of an implant through flat transesophageal ultrasound images contributes to a lengthy procedure, with a median total time of 88 minutes seen in a previous Edwards clinical trial of its Pascal platform.
Meanwhile, in one patient, clinicians reported using DeviceGuide to move into the left ventricle, optimize the position and orientation of the Pascal Ace, grasp the mitral valve’s leaflets and finally close the implant—all in under two and a half minutes.
“Structural heart procedures are among the fastest-growing areas in cardiology, and also among the most complex,” said Mark Stoffels, leader of Philips’ image-guided therapy business. “By assisting physicians with real-time visualization and navigation inside the beating heart, DeviceGuide helps them manage that complexity, and perform procedures more confidently, with the ultimate goal to treat more patients effectively.”
Related
This week also saw Philips launch its newly redesigned IntraSight Plus system for coronary procedures, following regulatory green lights in the U.S. and Europe.
The software combines real-time device visualization with intravascular ultrasound and angiography, plus fractional flow reserve measures, into one screen so clinicians can observe the impacts of artery-blocking plaque deposits as they navigate balloons and stents.