In a new study published in Cell titled, “Hyperinnervation inhibits organ-level regeneration in mammalian skin,” researchers from Harvard University reveal a method to fully regenerate skin by unblocking an embryonic healing mechanism that shuts off after birth. Demonstrated in mice, the study suggests a potential means to develop similar therapies in human patients.
“Essentially, we found a way to make wound healing outcome a lot better by learning how embryos do this so well,” said Ya-Chieh Hsu, PhD, professor of stem cell and regenerative biology at Harvard Stem Cell Institute and corresponding author of the study. “I’m excited because we pushed the needle in a really important direction. When we have a wound, most skin cell types cannot regenerate and we get a scar. But now I think we’ve found a way to change that, so that many cell types can regenerate and we don’t get a scar.”
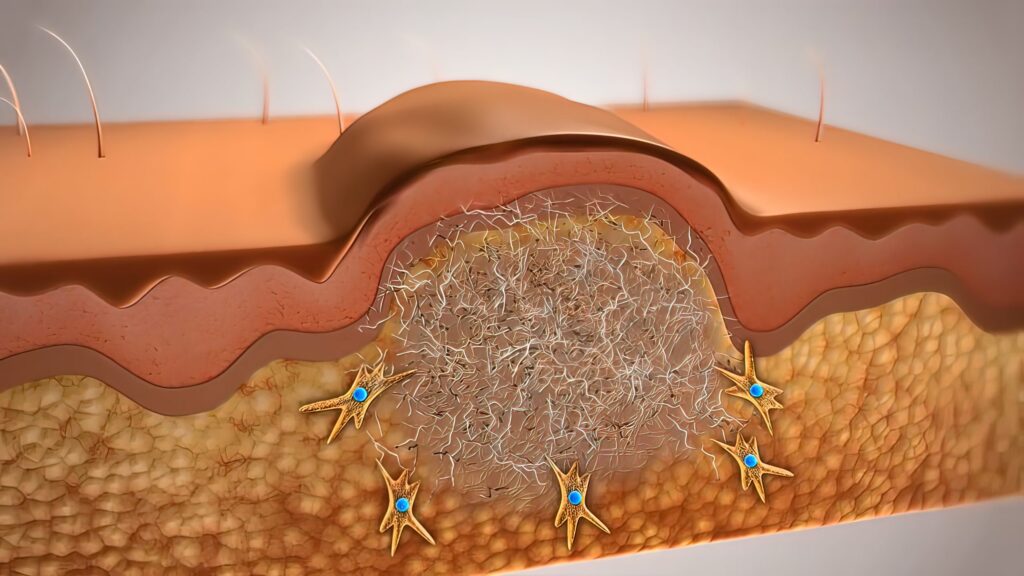
After injury, epidermal stem cells reseal the surface and fibroblasts deposit dense collagen scar tissue. However, skin also contains an array of additional cells that fail to regenerate leading to scarred skin. These cells include hair follicles, vascular and lymphatic vessels, sweat glands, pigment cells, immune cells, fat cells, and nerves.
After injury, embryonic skin restores all cell types but this ability fades quickly after birth. The study reveals the molecular mechanisms behind this switch and how to trigger reactivation.
“Our findings suggest that some organs retain an inherent regenerative potential that is simply held in check—and that removing this block may be sufficient to allow regeneration to occur,” Hsu said. “In other words, regeneration may not need to be built anew, but simply set free.”
The new findings culminated five years of research by lead author Hannah Tam, PhD, a graduate of the Harvard Kenneth C. Griffin Graduate School of Arts and Sciences in the biological and biomedical sciences program at Harvard Medical School.
To investigate wound healing, Tam compared how the organ regenerated in embryonic mice and postnatal mice at several timepoints. The team found that the ability to regenerate skin steadily decreased in the days after birth. The most dramatic shift occurred within a tight eight day window, from three days before birth to five days after birth.
In mice wounded three days before birth, the skin regenerated diverse cell types and closely resembled unwounded skin. However, wound sites five days after birth were covered by epithelial cells, packed with collagen scar tissue and abnormally dense nerve fibers and immune cells. Many other skin cell types failed to regrow.
The authors found that postnatal wound sites became densely packed with nerves. This “hyperinnervation” occurs because fibroblasts in postnatal wounds upregulate the gene Cxcl12, which recruits excessive nerves to the injury site and impairs the regrowth of other skin cell types.
Depleting Cxcl12 in wounds in postnatal mice curtailed hyperinnervation and the skin regrew diverse cell types. Blocking local nerve signaling with botulinum toxin A (Botox) produced similar effects.
Tam said the team “hit a wall” midway through their research because they assumed the regeneration process somehow involved immune cells. They soon discovered that the signaling behind the hyperinnervation could be switched off to restore full skin regeneration.
Tam explained that the block is through the fibroblast-nerve interaction. “The relationship between those two different cell types has not been the focus in wound healing studies,” she said. “I feel that this is very helpful to the field, because now we can really consider these two as actual communicators.”
Before the study, Hsu expected that the key to wound healing would be recreating a series of “regeneration-promoting factors” to mimic embryonic healing. The solution turned out to be simpler.
“I didn’t think that we’d have to retract a brake, which actually is good news—it’s a lot easier,” Tam said. “It gives me hope that this might be applicable to improving wound healing in humans.”