Gene therapy has entered a complicated phase. Over the past several years, the field has delivered remarkable scientific breakthroughs alongside sobering challenges—clinical safety concerns, manufacturing bottlenecks, and treatments priced in the millions of dollars. Some biotechnology companies have scaled back programs, while investors have grown cautious about the pace of commercial progress.
Despite these growing pains, gene therapy remains one of the most promising strategies for treating genetic diseases. Beneath the headlines, researchers and technology developers are quietly transforming the infrastructure that makes these therapies possible.
At the center of that transformation is the adeno-associated virus (AAV), a tiny viral vector that has become the dominant platform for directly delivering therapeutic genes into human tissues. AAV-based therapies have already shown encouraging clinical results in diseases ranging from inherited retinal disorders to neurological syndromes.
But the rapid expansion of AAV-based therapies has also exposed important limitations. Manufacturing these complex biological products at scale is difficult, ensuring consistent product quality requires advanced analytical tools, and naturally occurring AAV variants often lack the tissue-specificity needed to efficiently deliver genes without high doses.
Across the cell and gene therapy ecosystem, scientists and companies are now addressing these challenges through innovations in bioprocessing, vector engineering, and computational design. These advances could determine whether gene therapy evolves into a sustainable therapeutic platform capable of reaching larger numbers of patients.
Building scalable workflows
Manufacturing viral vectors remains one of the most technically demanding aspects of gene therapy development. Producing AAVs requires complex biological systems, typically involving cultured mammalian cells that generate the virus after being supplied with the necessary genetic components.
As gene therapy programs move from laboratory research into clinical trials, production requirements expand rapidly. Processes that work well at a small scale often struggle to maintain efficiency and consistency when scaled to large bioreactors.
One crucial challenge involves balancing upstream production—where the virus is generated—with downstream processing—where the vector is purified and formulated for clinical use.
Pouria Motevalian, PhD, director, CMC development, pharma services, at Thermo Fisher Scientific, says that successful scale-up depends on developing a detailed scientific understanding of the process early in development.
“Effective downstream scale-up begins with the establishment of reliable scale-down models,” Motevalian explains. “These models are then used to define the design space—the scientifically established operating region in which critical process parameters and material attributes can vary without compromising product quality.”
This concept of design space comes from quality by design, a development philosophy that emphasizes understanding how manufacturing variables influence product attributes. By mapping these relationships early, developers can design processes that remain robust as they scale.
“Parameters are defined not only by target setpoints, but also by proven operating ranges,” Motevalian says. “As a result, facility, equipment, and material constraints can be accommodated during scale-up without compromising process performance or product quality.”
Such flexibility becomes increasingly important as upstream technologies improve. New cell-culture strategies, optimized media formulations, and refined production platforms are allowing developers to generate higher quantities of AAV particles. But these gains can introduce downstream bottlenecks if purification systems are not prepared to handle the additional load.
“As upstream yields increase, downstream bottlenecks related to loading capacity, filtration throughput, buffer demand, and facility fit can be proactively addressed while maintaining recovery and product quality,” Motevalian notes.
Automation and high-throughput development platforms are also helping accelerate process optimization. These technologies enable rapid testing of multiple process conditions, allowing researchers to evaluate purification strategies and more efficiently optimize workflows.
Transforming viral-vector analytics
Manufacturing improvements alone cannot support large-scale gene therapy production. Developers must also demonstrate that each batch of viral vectors meets strict quality standards, which places heavy demands on analytical technologies. “The latest innovation in viral-vector analytics is centered on faster turnaround, lower sample consumption, and more multiplexed methods,” Motevalian says.
Traditional techniques, such as analytical ultracentrifugation (AUC), remain essential for evaluating vector composition, but emerging complementary technologies are providing additional insights. “Increasing emphasis is being placed on rapid orthogonal tools, such as mass photometry, as complements to gold-standard methods, such as AUC,” Motevalian explains. “At-line approaches, such as size-exclusion chromatography with multi-angle light scattering, also enable quicker assessment of vector-genome and capsid titers.” These methods allow developers to monitor vector quality more quickly during development, reducing delays and enabling faster decision-making.
Meanwhile, molecular techniques are becoming more efficient. Multiplex droplet digital PCR (ddPCR) assays, for example, allow researchers to simultaneously analyze several genomic regions. “Multiplex ddPCR strategies are expanding analytical efficiency by enabling simultaneous interrogation of multiple genome regions and selected impurities,” Motevalian says.
Another emerging trend is the adoption of multi-attribute analytical methods based on liquid chromatography–mass spectrometry (LC-MS). These approaches allow several critical quality attributes to be monitored within a single analytical workflow.
The ability to track multiple parameters simultaneously strengthens process understanding and supports regulatory comparability assessments.
Engineering better vectors
Although manufacturing and analytics are improving rapidly, gene-therapy developers must still contend with the biological limitations of naturally occurring AAV vectors. Many widely used AAV serotypes were discovered decades ago and did not originally evolve for precision gene delivery in humans. As a result, they often lack the targeting specificity needed for efficient therapy.
“There are three main roadblocks, and they are all interdependent: delivery, safety, and the cost of scaling up manufacturing,” says Amos Gutnick, PhD, associate director of product development at PackGene. “Natural AAVs, like AAV9 or AAV2, still struggle with precision, so patients currently require massive doses in order to benefit from a therapeutic effect.”
Those high doses can “introduce serious safety risks, like liver toxicity and adverse immune reactions,” Gutnick notes. “They also drive production costs through the roof, often making the program commercially untenable.”
To overcome these obstacles, developers are working simultaneously on improving vector design and optimizing manufacturing efficiency. “At PackGene, our company motto is literally: ‘Make Gene Therapy Affordable,’” Gutnick says. Part of that effort involves improving bioprocessing platforms to generate higher yields and better quality vectors.
“On the manufacturing end, we’re constantly innovating our bioprocessing platforms to boost overall yields and purity,” Gutnick explains. “These innovations empower us to manufacture GMP-grade plasmids at record-breaking low costs and maximize the percentage of full, functional AAV capsids at every scale.”
At the same time, advances in capsid engineering are enabling more precise gene delivery. “On the design side, we work to engineer novel AAV capsids that are highly specific to the target tissue with the goal of increasing safety and drastically lowering the therapeutic dose,” Gutnick says. “When you combine better manufacturing yields with much lower clinical doses, the economics completely change.” And, he adds, “that can mean the difference between an interesting paper and an actual, commercially viable gene therapy program.”
AI accelerates vector design
Artificial intelligence (AI) is increasingly shaping how new viral vectors are developed. Machine-learning algorithms can analyze large datasets describing how capsid sequences behave in biological systems and use those insights to design improved variants.
As Gutnick says, “AI is a cornerstone of our π-Icosa Capsid Engineering Platform.” These models are trained to optimize both biological performance and manufacturability. “Biologically, the AI helps us design novel AAV capsids with incredible tissue targeting while actively detargeting the liver,” Gutnick explains.
Avoiding liver accumulation is particularly important because the liver often receives a large share of systemically delivered vectors and can become a source of toxicity. Equally important is ensuring that engineered capsids can be produced efficiently. “But just as importantly, we use AI to design for high manufacturability from day one,” Gutnick says.
Historically, some promising vectors discovered in the laboratory proved difficult to manufacture at scale. Designing with production in mind helps reduce that risk.
Faster effective-vector discovery
Identifying the right AAV capsid for a particular therapeutic application can take years. To accelerate this process, researchers are developing tools that allow large numbers of vector variants to be tested simultaneously.
As Gutnick says, “One of the biggest bottlenecks for translational researchers is simply the time and money it takes to find the right vector.”
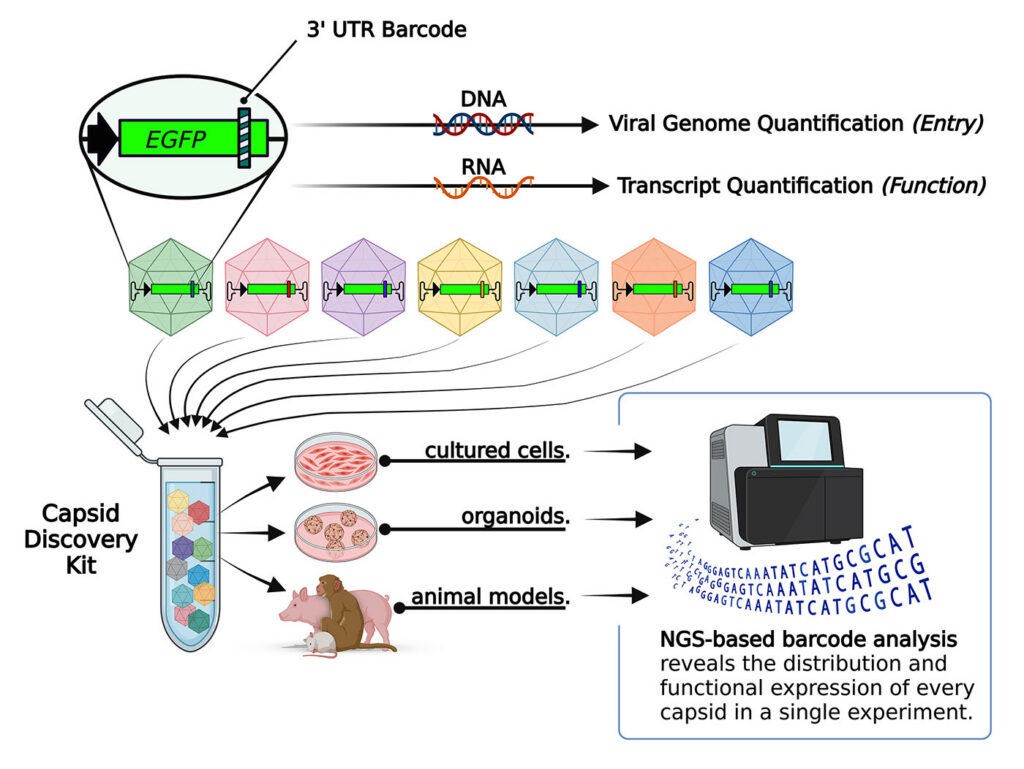
To help address that challenge, PackGene collaborated with the Children’s Medical Research Institute to develop capsid-discovery kits that enable multiplex screening experiments. “This partner used this off-the-shelf kit to run high-throughput, multiplexed in vivo screens on dozens of capsids all at once,” Gutnick explains.
The screening system uses next-generation sequencing (NGS) to track both where vectors travel in the body and whether they successfully express their genetic payload. “By using dual NGS readouts to track both where the AAV went and if it actually worked, they were able to quickly pinpoint a lead capsid that hit their target tissue perfectly while avoiding off-target areas,” Gutnick says. “By giving researchers access to these kits, we’re helping them cut months—even years—off their discovery timelines.”
Platforms streamline production
Although improvements in vector design and analytics are essential, the infrastructure needed to manufacture gene therapies at scale is also evolving.
For example, Catalent developed an AAV production platform designed to accelerate the path from gene discovery to clinical manufacturing. The approach relies on standardized suspension HEK293 cell culture systems and integrated supply chains that combine plasmid production, process development, and manufacturing services.
Charles River Laboratories introduced its nAAVigation vector platform to streamline viral-vector development. Built around a high-productivity HEK293 suspension cell line and optimized upstream and downstream processes, the platform is intended to reduce development timelines while enabling scalable production.
Synthetic biology company Asimov is exploring another strategy through its AAV Edge Stable Producer System. Rather than relying on transient transfection for each production run, the system uses engineered producer cell lines in which viral genes are integrated directly into the genome. These stable cell lines can produce AAV vectors more consistently and might reduce manufacturing costs by eliminating the need for multiple plasmids.
Meanwhile, Andelyn Biosciences is applying its AAV Curator manufacturing platform to support gene-therapy programs targeting rare diseases. The company is collaborating with the Drake Rayden Foundation and researchers at the University of Texas Southwestern to manufacture clinical-grade vectors for a potential therapy aimed at nonketotic hyperglycinemia, a severe inherited metabolic disorder.
Toward a sustainable ecosystem
The rapid evolution of AAV technology reflects a broader shift in gene therapy from experimental science toward industrial-scale medicine.
Manufacturing innovations are enabling more efficient production of viral vectors. Advanced analytics are providing deeper insight into vector quality and performance. And computational approaches are unlocking new possibilities for designing safer, more precise delivery systems.
Together, these advances suggest that the next chapter of gene therapy will be defined not only by scientific discovery but also by the ability to manufacture and deliver treatments reliably and at scale. If those efforts succeed, AAV vectors could help transform the treatment of genetic disease—turning once-experimental therapies into widely accessible medicines capable of changing patients’ lives.