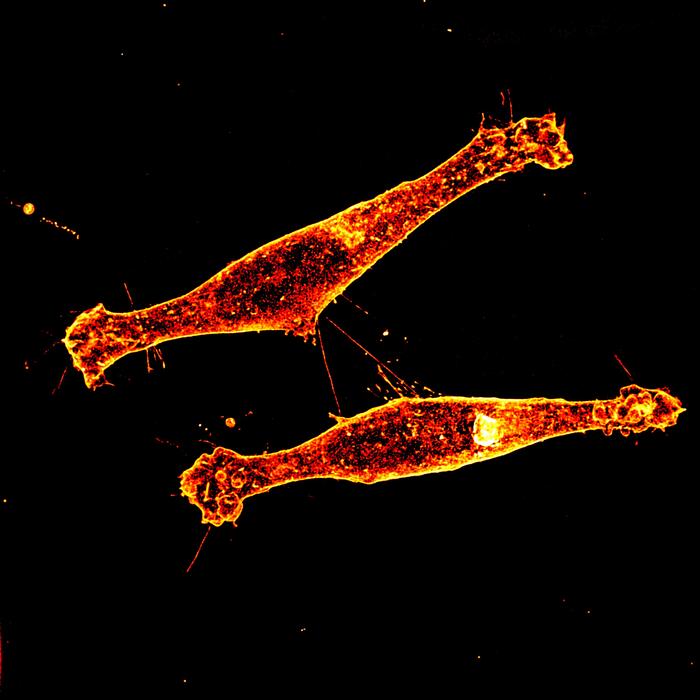
![DO NOT REUSE Tunneling nanotubes connect Rhes expressing striatal neuronal cells [Emaad Mirza, Florida Atlantic University]](https://www.genengnews.com/wp-content/uploads/2026/03/low-res-nanotubes-696x696.jpeg)
Huntington’s disease (HD) is a devastating brain disorder that slowly robs people of movement, memory and personality. It is caused by a toxic, mutant Huntingtin (mHTT) protein that builds in brain cells and ultimately kills them. For years, scientists have known that this harmful protein doesn’t stay put—it spreads from one brain cell to another. However, exactly how that spread happens and how to stop it has remained a mystery.
In a major breakthrough, researchers from Florida Atlantic University and collaborators have identified a previously unknown cellular pathway that allows brain cells to pass toxic material directly to their neighbors through tiny, tube-like structures called “tunneling nanotubes.”
The new research reveals that a protein called Rhes—already known to play a key role in Huntington’s disease—partners with a bicarbonate transporter called SLC4A7, which is a protein best known for helping cells regulate their internal acidity. Together, these two proteins help to build tunneling nanotubes, creating highways that allow toxic Huntingtin protein to move from one neuron to another.
Importantly, the study showed that disrupting this pathway in mice dramatically reduced the spread of the disease-causing protein in the brain. The findings could point to therapeutic strategies for Huntington’s disease, as well as other neurodegenerative disorders and potentially cancer.
“This work fundamentally changes how we think about disease progression in Huntington’s,” said Srinivasa Subramaniam, PhD, senior author, associate professor in the department of chemistry and biochemistry within FAU’s Charles E. Schmidt College of Science, and a member of FAU’s Stiles-Nicholson Brain Institute, David and Lynn Nicholson Center for Neuroscience Research, and the Center for Molecular Biology and Biotechnology.
“We’ve known that neurons somehow pass toxic proteins to one another, but now we can see the machinery that makes that possible. By identifying SLC4A7 as a key partner of Rhes, we’ve uncovered a new and potentially druggable target to stop that spread at its source.” Subramaniam is senior and corresponding author of the team’s published paper in Science Advances, titled “Membrane-associated Rhes-Slc4a7 complex orchestrates tunneling nanotube formation and mutant Huntingtin spread.”
Communication between cells in the brain is essential for maintaining neural function and responding to injury or disease, the authors wrote. “One emerging mode of such communication is through tunneling nanotubes (TNTs)—thin, actin-based membranous structures that form direct cytoplasmic bridges between cells.” TNTs mediate intercellular transfer of proteins, including the pathogenic mutant Huntingtin protein in Huntington disease.
Unlike chemical signals that diffuse through space, nanotubes allow cells to share proteins and other materials by hand-delivery. While this kind of sharing may sometimes help healthy cells respond to stress or injury, it can also become dangerous when it spreads harmful proteins, such as the mutant Huntingtin protein. And while the activity of TNTs is of particular interest in the context of neurodegeneration, the authors continued, “… the precise molecular regulators of TNT formation and their physiological relevance in the brain remain poorly understood.”
The team had previously identified Rhes as a key regulator of TNT formation and mHTT. Transmission, however, they noted, “… the molecular components underlying this process remained unknown.” Using advanced protein-mapping techniques, the researchers have now discovered that Rhes physically binds to SLC4A7, in intracellular pH sensor, at the cell membrane. “When this partnership forms, it triggers changes inside the cell that promote the growth of nanotubes. When the team blocked SLC4A7—either genetically or with drugs—the nanotubes failed to form, and the toxic Huntingtin protein was largely unable to spread. “Functional studies revealed that small interfering RNA–mediated depletion or pharmacological inhibition of Slc4a7 significantly reduced Rhes-induced TNT formation and suppressed mHTT intercellular transfer,” they wrote.
Significantly, this effect wasn’t just seen in isolated cells. In mouse models of Huntington’s disease, mice lacking SLC4A7 showed a dramatic reduction in the transfer of toxic protein between neurons in the brain’s striatum, the region most affected in the disease. “Slc4a7 knock-out mice showed markedly reduced cell-to-cell transmission of mHTT in the striatum in vivo,” write the authors. This suggests that interfering with this newly discovered pathway could slow the progression of Huntington’s disease by containing the damage before it spreads.
The implications from this study extend far beyond Huntington’s disease. Tunneling nanotubes have been implicated in other neurodegenerative disorders, including conditions involving tau protein, as well as in cancer, where tumor cells use similar structures to share signals, energy and even drug resistance. “A growing body of evidence supports a role for TNTs in cancer progression,” the team noted. Because both Rhes and SLC4A7 are involved in fundamental cellular processes, the newly identified pathway may represent a common mechanism underlying the spread of damage in many diseases. “Our findings that Rhes and Slc4a7 promote TNT formation suggest a broader role for these proteins in the pathological spread of misfolded proteins in both neurodegenerative diseases and cancer.”
“This research shines a spotlight on an entirely new way cells communicate in health and disease,” said Randy Blakely, PhD, executive director of the FAU Stiles-Nicholson Brain Institute, the David J.S. Nicholson Distinguished Professor in Neuroscience, and a professor of biomedical science in the FAU Charles E. Schmidt College of Medicine. “By learning how harmful proteins physically move from cell to cell, we gain powerful new leverage points for therapy. The idea that we could slow or even halt disease progression by blocking these microscopic tunnels opens an exciting frontier for treating not only Huntington’s disease, but a wide range of neurological disorders and cancers in the future.”
As scientists continue to unravel how cells share information—and how that sharing can go wrong—this discovery offers new hope that stopping disease may one day be as simple as closing the door between cells. “Together, the involvement of Rhes in both mHTT and tau pathologies, along with its interaction with Slc4a7, suggests a shared mechanism facilitating protein spread and neurotoxicity across multiple neurodegenerative diseases,” the scientists concluded. “Therefore, targeting the Rhes-Slc4a7 axis could represent a novel therapeutic strategy to disrupt TNT-mediated intercellular communication and limit the dissemination of pathogenic proteins in neurodegeneration and cancer.”
Huntington’s disease is a rare, inherited brain disorder that affects about three to seven people per 100,000 worldwide and strikes men and women equally. Symptoms usually appear between the ages of 30 and 50 years and worsen over time, causing uncontrolled movements, cognitive decline, and serious psychiatric symptoms. Each child of an affected parent has a 50% chance of inheriting the disease. There is no cure, and current treatments only help manage symptoms without stopping progression. After symptoms begin, people typically live 10 to 20 years, often facing increasing disability and loss of independence.