The Data Multiplier Effect: When Expertise, Monitoring, Systems, and AI Align
By Amir Malka, CEO, Bioforum The Data Masters
Clinical research is evolving rapidly. Trials are more complex, data sources keep expanding, and regulators expect greater transparency and reproducibility. In parallel, AI is transforming how data is collected, curated, and analyzed.
The question for sponsors is no longer how to gather more data, but how to turn it into reliable, audit-ready insight. The answer is the alignment of four forces: specialized expertise, data-led centralized monitoring, interoperable systems, and responsible AI.
When these come together, data stops moving in parallel streams and begins to converge, revealing relationships that were not visible before. This dynamic is what we at Bioforum call the data multiplier effect. It’s beginning to redefine how sponsors approach clinical development, a shift we’re seeing take shape in four key areas.
1. Specialized Data Partnerships
Across the industry, a new model is gaining ground as broad outsourcing strategies reveal their limits. Specialized partners with deep expertise in biometrics, pharmacovigilance, and data management bring focus and discipline to the data process and help ensure each dataset is reliable and ready for review.
Real-world data integration shows why this matters. Sponsors can pull data from registries, EMRs, and decentralized platforms, yet without experienced oversight those sources remain fragmented and difficult to validate. The challenge is making diverse datasets coherent and submission-ready.
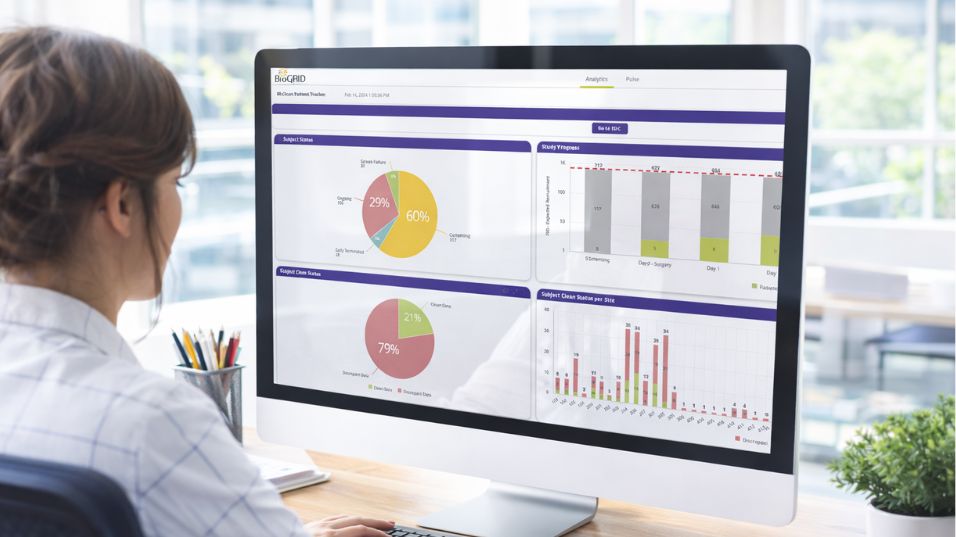
A recent Bioforum Phase II oncology engagement shows how specialized biometrics can improve data quality and operational efficiency. The study drew on several data streams, integrated through BioGRID, Bioforum’s visual analytics platform designed to unify clinical data sources for real-time review and oversight.
With biometrics-led oversight, reconciliation and data review were streamlined, reducing data-query turnaround time by roughly 33%, completing interim reviews about 25% faster, and achieving database lock about ten days ahead of plan with strengthened audit readiness.
2. Data-Led Centralized Monitoring
With rising trial complexity and expanding data streams, data-led centralized monitoring provides continuous, analytical oversight that supports quality, safety, and execution across the trial lifecycle.
When centralized monitoring is led by biometrics and data experts, continuous data review, cross-site trend analysis, and early detection of quality or safety signals become control functions that guide decisions in real time.
By converting integrated data into actionable signals, data-led centralized monitoring enables proactive intervention, strengthens RBQM frameworks, and reduces reliance on reactive, manual processes. The result is earlier risk detection, stronger data integrity, and greater confidence across sponsors, sites, and regulators.
3. Connected Technology Ecosystems
Sponsors are increasingly pairing core platforms such as Medidata, Veeva, and Viedoc with complementary solutions that improve automation, validation, and regulatory readiness. Bioforum extends this approach with proprietary tools that close integration gaps:
- BioGRID integrates disparate data sources into harmonized, analytics-ready environments.
- JetConvert automates data conversion and validation, reducing manual burden and the risk of error.
- Validify adds a critical validation layer, embedding integrity checks at every stage.
In a Bioforum rare disease study with niche external datasets and variable lab formats, this combination improved data readiness, including reconciliation cycles about 30% faster and manual transformation effort reduced around 40%.
4. AI as an Embedded Partner
AI is reshaping how studies are designed, monitored, and analyzed, though results remain mixed. The goal is not to deploy AI everywhere, but to embed it strategically within validated processes under expert oversight, strengthening execution without replacing human judgment. High-impact applications tend to be specific and measurable:
- Faster data cleaning and reconciliation by flagging anomalies and inconsistencies earlier
- Earlier operational and data-quality signals to support risk-based monitoring across sites or patients
- Support for regulated workflows such as SDTM value and variable mapping through in-workflow recommendations under human oversight (in JetConvert)
- Stronger validation and audit readiness through AI-assisted risk identification, as implemented in Validify
- Faster interrogation of complex datasets through natural-language interaction and automated visualizations, including BioGRID’s AI assistant, BRIAN
At Bioforum, we view AI as a reinforcing layer within the biometrics process that connects design, data, and decision-making more intelligently.
The Multiplier Effect
Each of these areas, specialized expertise, data-led centralized monitoring, connected systems and the embedded use of AI, delivers measurable value on its own. But when they work together, their impact multiplies:
- Trials move faster without compromising quality
- Data becomes interoperable, reliable, and ready for regulatory review
- Automation reduces manual work and improves accuracy, consistency, and efficiency
- Confidence grows among sponsors, sites, and regulators.
This is the data multiplier effect in action: when expertise, technology, and intelligence converge, the value of data compounds, advancing science and improving lives.
About the Author
Amir Malka, Adv. is the Co-Founder and CEO of Bioforum The Data Masters, a biometrics-focused CRO. With 25+ years in life sciences, his vision of aligning operational excellence and technology has shaped Bioforum’s evolution into a global, data-driven CRO known for disciplined execution.
Amir’s leadership and entrepreneurial impact have been widely recognized. He was named among PharmaVOICE 100 in 2021, and Bioforum has been nominated for three consecutive years as a finalist for Best Specialty CRO at the Scrip Awards.