Intestinal ischemia/reperfusion (I/R) injury represents a significant clinical challenge characterized by the temporary cessation and subsequent restoration of blood flow to the intestines, leading to severe tissue damage. This pathological process is intricately linked with conditions ranging from acute mesenteric ischemia to complications during surgeries involving blood flow interruption. The resultant cellular damage and inflammation often precipitate systemic responses, contributing to multiple organ dysfunction and a high mortality rate. Despite its critical impact, the molecular underpinnings governing intestinal I/R injury remain incompletely understood, mandating rigorous research to uncover therapeutic targets that can mitigate this devastating condition.
In a groundbreaking study recently published in the Chinese Medical Journal, Professor Xiaofeng Tian and colleagues from Dalian Medical University illuminate the pivotal role of the growth arrest and DNA damage-inducible beta (GADD45B) in exacerbating apoptosis during intestinal I/R injury. This research elucidates a novel epigenetic axis involving GADD45B, ten-eleven translocation methylcytosine dioxygenase 1 (TET1), and mammalian sterile 20-like kinase 1 (MST1), unveiling the molecular choreography that intensifies apoptotic signaling through the Hippo pathway following ischemic insult.
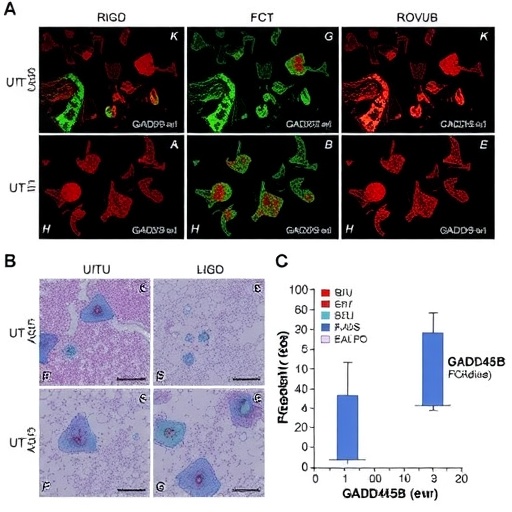
GADD45B, a multifunctional stress sensor, orchestrates a spectrum of cellular responses encompassing DNA repair, apoptosis, cell cycle arrest, and inflammatory modulation. Prior transcriptomic analyses have flagged GADD45B as markedly upregulated in human intestinal tissue subjected to I/R injury, an observation further supported by murine model studies. This robust upregulation hinted at a potential causative role for GADD45B in the pathophysiology of ischemia-reperfusion damage, prompting a comprehensive exploration of its mechanistic contributions.
Employing both in vitro models using hypoxia/reoxygenation (H/R)-induced Caco-2 cells and in vivo murine I/R paradigms, the investigation demonstrated a significant elevation of GADD45B expression post-injury. Functional assays revealed that targeted knockdown of GADD45B curtailed apoptosis and mitigated tissue damage not only within the intestine but also in remote organs affected by systemic inflammatory responses. Contrariwise, forced overexpression of GADD45B heightened susceptibility to cell death and aggravated intestinal injury, underscoring its pathogenic potency.
Delving deeper, the research team applied transcriptomic KEGG pathway enrichment analysis, which spotlighted the Hippo signaling cascade as a downstream effector of GADD45B. MST1, a core kinase within the Hippo pathway known to influence cell proliferation and apoptosis, emerged as a critical target. The study probed the epigenetic regulation of MST1, revealing that GADD45B modulates its expression via DNA demethylation in the promoter region, a process conventionally governed by TET family demethylases.
TET1, recognized for catalyzing the oxidation of 5-methylcytosine to enable active DNA demethylation, was found to exhibit expression patterns concordant with GADD45B following intestinal I/R injury. Chromatin immunoprecipitation (ChIP), methylation-specific PCR (MSP), and co-immunoprecipitation (Co-IP) experiments substantiated a physical and functional interaction between GADD45B and TET1. This complex facilitated region-specific DNA demethylation, thereby enhancing MST1 transcription and amplifying the activation of the Hippo pathway.
The activation of the Hippo pathway by MST1 culminates in the phosphorylation and nuclear translocation of downstream effectors, which orchestrate transcriptional programs favoring apoptosis. Consequently, the GADD45B-TET1-MST1 axis intensifies cell death mechanisms in intestinal epithelial cells subjected to ischemia and reperfusion stress, exacerbating tissue damage and compromising intestinal barrier integrity. This newly delineated epigenetic mechanism enriches our understanding of ischemia-induced apoptosis, bridging molecular biology with pathophysiological outcomes.
These insights carry profound therapeutic implications. By targeting components of the GADD45B/TET1/MST1 signaling axis, it may be possible to attenuate the detrimental apoptotic responses triggered during I/R injury, preserving intestinal function and preventing subsequent systemic complications. Pharmacological interventions aimed at disrupting GADD45B interactions or modulating epigenetic regulators like TET1 could herald innovative treatments for ischemic intestinal diseases.
The significance of this work extends beyond intestinal pathology, as similar epigenetic regulatory mechanisms may operate in other ischemic tissues, including the heart and brain. Moreover, GADD45B’s established roles in DNA damage response and inflammation suggest a broader influence in diverse stress-related diseases, positioning it as a versatile target for future drug development.
Professor Xiaofeng Tian’s team has pioneered a compelling narrative that reconceptualizes the molecular landscape of intestinal I/R injury, highlighting how epigenetic modulation governs cellular fate decisions. This study not only expands the scientific community’s comprehension of ischemic injury but also exemplifies the intricate interplay between genetic regulation and disease phenotypes.
Collectively, these findings endorse GADD45B as a central modulator orchestrating the epigenetic activation of MST1 via TET1-mediated demethylation, ultimately triggering the Hippo pathway and heightening apoptotic injury in ischemic intestines. Continued exploration of this pathway promises to yield novel diagnostic markers and therapeutic avenues, potentially transforming clinical management strategies for patients afflicted by intestinal ischemia and reperfusion injuries.
As the field advances, integrating epigenetic profiling with functional studies will be crucial in unraveling the complex regulatory networks driving ischemic injury responses. The work by Professor Tian and colleagues lays a robust foundation for such integrative research, offering a blueprint for future investigations that might unlock new paradigms in ischemia biology and treatment.
This discovery shines a spotlight on the transformative power of epigenetic mechanisms in disease progression and emphasizes the necessity of multidisciplinary approaches combining molecular genetics, cellular biology, and clinical insights to combat ischemia-reperfusion pathology effectively.
Subject of Research: Animals
Article Title: GADD45B promotes apoptosis in intestinal ischemia/reperfusion through DNA demethylation of MST1/Hippo
News Publication Date: 9-Apr-2026
Web References:
https://journals.lww.com/cmj/fulltext/9900/gadd45b_promotes_apoptosis_in_intestinal.2014.aspx
http://dx.doi.org/10.1097/CM9.0000000000004050
References:
DOI: 10.1097/CM9.0000000000004050
Image Credits:
Professor Xiaofeng Tian from Dalian Medical University
Keywords:
Cell apoptosis, Cell biology, Molecular biology, Gene expression, Epigenetics, Gastrointestinal disorders, Ischemia, Signal transduction, Biochemistry
Tags: apoptosis modulation by GADD45Bcell cycle arrest in ischemic injuryDNA damage response in intestinal cellsepigenetic regulation in intestinal injuryGADD45B role in apoptosisHippo pathway in ischemiainflammation in intestinal ischemiaintestinal ischemia-reperfusion injurymolecular mechanisms of intestinal I/R injurysystemic effects of intestinal ischemia reperfusionTET1 and MST1 signaling pathwaytherapeutic targets for ischemic bowel disease