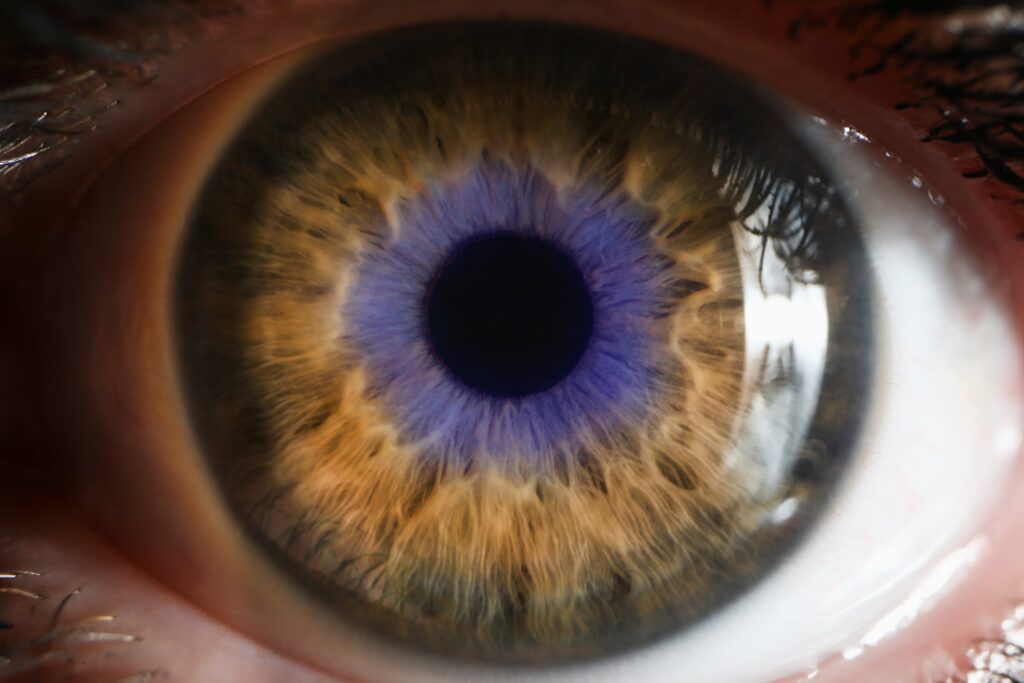
Gene therapy biotech Ray Therapeutics has announced the closing of a $125 million series B financing to support the company’s vision restoration candidates for patients with retinal degeneration.
The oversubscribed round was led by Janus Henderson Investors, with participation from new investors Adage Capital Management, Franklin Templeton, Invus and Marshall Wace, alongside follow-on support from existing backers 4BIO Capital, Deerfield Management, MRL Ventures Fund, Norwest, Novo Holdings and Platanus, according to an April 21 release.
The funding follows the FDA’s move to grant its Regenerative Medicine Advanced Therapy (RMAT) designation to Ray’s lead candidate, RTx-015, which is designed to treat retinitis pigmentosa (RP).
The RMAT designation is intended for therapies targeting serious conditions with unmet need and offers benefits such as intensive FDA guidance, a rolling review and the potential for a priority review.
In a statement, Ray CEO and co-founder Paul Bresge called the round a “decisive endorsement” of the company’s platform, underscoring “the clinical and regulatory momentum we have achieved to date.”
RP, a genetic disorder that causes progressive vision loss due to photoreceptor cell breakdown, has been a particularly active area within biopharma as of late.
Ocugen CEO Shankar Musunuri, Ph.D., told Fierce Biotech earlier this month that he hopes the company can bring retinal treatments to a broader patient population. Its lead asset is currently in a phase 3 trial for RP, and Musunuri said the company plans to begin a rolling FDA submission this year. He described the study as the “largest orphan gene therapy trial in the world.”
Related
It hasn’t all been good news about treating RP. Earlier this week, Johnson & Johnson sold an RP asset back to MeiraGTx for $25 million upfront after it failed a phase 3 trial last year. Johnson & Johnson had originally acquired the candidate in a deal worth up to $415 million in 2023.
Despite the setback and uncertainty around FDA leadership, MeiraGTx said it hopes to pursue a rapid filing in the U.S. and EU, with a potential 2027 launch.
Agustin Mohedas, portfolio manager and research analyst at Janus Henderson Investors, expressed confidence that Ray could succeed where J&J failed. “RayTx’s platform is not just an incremental improvement; it is a first- and best-in-class technology with the potential to restore independence to patients and set a new standard of care in this patient population,” he said.