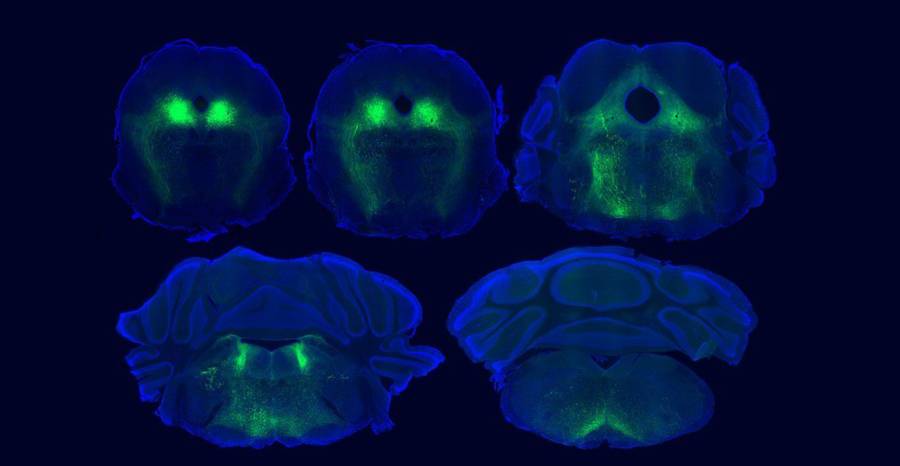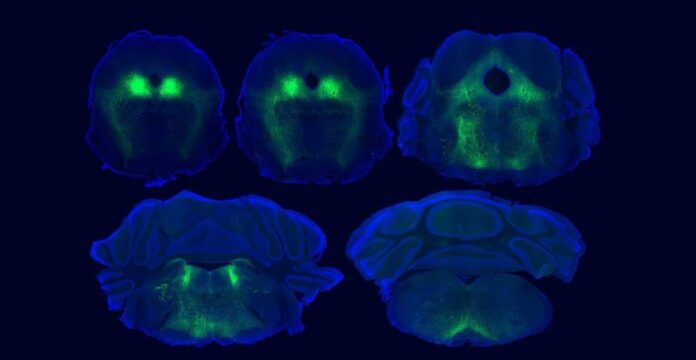
Though the placebo effect is a well documented phenomenon, the neurological mechanisms that underlie the process are still not fully understood. Now scientists from multiple institutions led by a team at the University of California San Diego (UCSD) have pinpointed the brain circuitry in mice that they believe is responsible for placebo pain relief. Details of their findings are published in a new paper in the journal Neuron. In it, they describe brain regions that support placebo effects and highlight sites where endogenous opioid neuropeptides send signals that are important for placebo pain relief.
The paper is titled “Top-down control of the descending pain modulatory system drives multimodal placebo analgesia.” According to the team, theirs is the first study to establish placebo mechanisms by adapting a protocol used for humans to work in mice. Working alongside labs at the University of Pennsylvania, University of California Irvine, and elsewhere, the UCSD team detected activity in parts of the mouse brain that correspond to those previously implicated in human studies. Furthermore, by precisely mapping neural pathways and brain activity in the mice, the team identified essential roles for neural circuits that link the cortex to the brainstem and spinal cord during placebo pain relief.
They also found that training mice to exhibit a placebo effect with one type of pain results in relief from several different types of pain including pain from injuries. That is particularly notable because it has “direct implications for how placebo training in humans might be used to produce resilience to future pain that results from injury,” explained Matthew Banghart, PhD, an associate professor in UCSD’s neurobiology department and lead author on the study. The findings also open a door to “expectancy-driven” placebo effects as a substitute for addictive painkillers, he noted, meaning that it might be possible to use placebo conditioning to train patients to build preemptive resilience to pain.
Full details of the findings and methods used are provided in the paper. In it, the teams explain that they used sensor technology and a light-activated drug developed in the Banghart lab to study the role of naturally-occurring opioid peptides in the brain. Specifically, they used the sensors to detect opioid peptide signaling in the ventrolateral periaqueductal gray (vlPAG) region, a known hub for pain signaling, during placebo trials. They then used the light-activated drug called photoactivatable naloxone, or PhNX, to establish that these opioid peptides actually drive pain relief in a manner similar to drugs like morphine. The light allowed the scientists control and timing of the opioid signaling interference. Using PhNX, they confirmed that both morphine-induced pain relief and placebo pain relief use the same opioid signaling pathway in the vlPAG region of the brain.
Essentially, “we trained a mouse brain to create its own broad-spectrum painkillers on demand, precisely where they are needed to treat pain, without the off-target effects of opioid-based painkillers,” said Janie Chang-Weinberg, a PhD student in the biological sciences graduate program at UCSD and one of the first authors on the study.
Future studies planned by the team will dig more deeply into how placebo learning unfolds in the brain and evaluate different placebo training strategies in mice with an eye towards developing protocols that readily translate to produce placebo pain resilience in people living with chronic pain.